Evolution of Antibody Humanization and Affinity Maturation
Sumana Sundaramurthy, Technical Account Manager, Sino Biological
Antibodies play a vital role in the immune system by providing protection against foreign invaders, such as bacteria and viruses. Produced by B cells, antibodies are protein molecules that can be harnessed to treat cancers, autoimmune disorders, and various infectious diseases. However, the human immune system may recognize modified therapeutic antibodies as foreign substances, reducing their efficacy. To overcome this challenge, the following two processes have been developed: antibody humanization and affinity maturation.
Antibody humanization and affinity maturation have revolutionized the biotech industry in the past 10–20 years, enabling the development of highly effective therapeutic antibodies that can be used to treat a wide range of diseases. Prior to antibody humanization and affinity maturation, therapeutic antibody development was a slow and challenging process, often reliant upon less effective nonhuman sources that lead to elicitation of unwanted immune responses and low binding affinities. Antibody humanization and affinity maturation have changed this paradigm by enabling the development of therapeutic antibodies that are more effective at treating diseases. By enhancing binding to the target molecule, increasing half-lives in the body, and improving immune cell recruitment, these processes have improved therapeutic antibody effectiveness while avoiding elicitation of an immune response in humans. Therefore, the advancement of antibody humanization and affinity maturation technology holds immense potential in facilitating the development and enhancing the quality of therapeutic antibodies.
Antibody Humanization
Antibody humanization is a crucial process in the development of therapeutic antibodies, which involves modifying nonhuman antibody molecules to make them more compatible with the human immune system. Although nonhuman antibodies, such as those of mice and rabbits, are commonly used in research to develop therapeutic antibodies, they can elicit an immune response when used as therapeutic agents in humans, reducing their efficacy.
To achieve humanization, the amino acid sequence of a nonhuman antibody molecule is modified to match that of a human antibody molecule. X-ray crystallography and nuclear magnetic resonance spectroscopy are used to identify the antigen-binding site, and then bioinformatics and computational modeling allow the prediction of the structural consequences of framework and complementarity-determining region (CDR) modifications. This process enables the creation of a molecule that can be used as an effective therapeutic agent in humans without causing an immune response. Overall, antibody humanization is critical in developing therapeutic antibodies, as it enables them to be used in humans with reduced immunogenicity, improving their efficacy in treating diseases (Fig. 1).
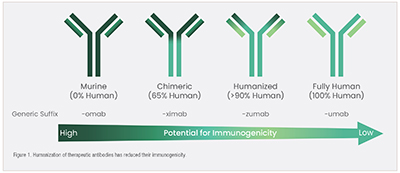
Sino Biological offers an antibody humanization service through its CDR grafting technology, providing a success rate of 100% and >95% sequence homology compared to human antibody frameworks. Our humanized antibody packages guarantee equal or higher affinity than any parental or chimeric antibody (Fig. 2).

Affinity Maturation
Affinity maturation is another integral process in the development of highly effective therapeutic antibodies. It involves creating mutations in the CDRs of an antibody, which results in structural changes that can facilitate optimal interactions with the antigen, thereby increasing its binding affinity, i.e., the strength of the interaction between the antibody and its target. The higher the affinity, the more effective the antibody is at binding to its target and eliciting a response. Traditionally, affinity maturation has involved multiple rounds of testing and modification to identify an amino acid sequence that will maximize the antibody’s affinity to the target.
Application of Artificial Intelligence
The use of artificial intelligence (AI) has revolutionized antibody humanization and affinity maturation in the biotech industry. By analyzing large datasets of antibody molecules, AI can identify specific amino acid sequences that are most likely to increase the binding affinity of the antibody to its target and be compatible with the human immune system. This has greatly reduced the time and cost involved in developing therapeutic antibodies, making the process more accessible to researchers and biotech companies. Consequently, there has been an increase in the number of therapeutic antibodies in development and a decrease in the time it takes for these antibodies to reach the market.
Sino Biological has partnered with Ainnocence to offer cutting edge AI-based antibody affinity maturation and denovo antibody design. Powered by Ainnocence’s SentinusAITM self-evolving AI search engine, this new platform effectively rank up to 1010 antibody sequences based on their predicted affinity toward one or more antigens. Then, Sino Biological’s high-throughput recombinant antibody development service can produce >1000 high-purity recombinant antibodies per project, with SentinusAITM validating the affinities of these antibodies.

Real-world Applications of Antibody Humanization and Affinity Maturation
Antibody humanization and affinity maturation have had a significant impact on the biotech industry, particularly in the development of monoclonal antibodies (mAbs) for cancer treatment. mAbs are designed to bind to specific molecules on cancer cells, triggering an immune response that leads to the destruction of the cells. Through humanization and affinity maturation, the development of mAbs with low immunogenicity, high specificity, and therapeutic activity has been facilitated, resulting in effective targeting of cancer cells. Another area influenced by antibody humanization and maturation has been the development of therapeutic antibodies to treat autoimmune diseases, such as rheumatoid arthritis and multiple sclerosis, which are caused by the immune system attacking the body’s own tissues. Humanized and affinity-matured antibodies have enabled the development of highly effective therapies that can block the immune response responsible for causing these diseases.
The humanization and affinity maturation of antibodies has also played a critical role in the development of highly effective vaccines that protect against a wide range of infectious diseases including viral infections like HIV, hepatitis C, and respiratory syncytial virus (RSV). In addition to their application in the development of therapies and vaccines, antibody humanization and affinity maturation have opened up new opportunities for biotech companies and researchers. By reducing immunogenicity and increasing specificity, these processes have made the development of new therapies and treatments possible.
Conclusion
Overall, antibody humanization and affinity maturation have revolutionized the biotech industry in the past two decades by enabling the development of highly effective therapeutic antibodies, vaccines, and other treatments. The use of AI in these processes has accelerated their development, making them more accessible to biotech companies and researchers. As these technologies continue to evolve, they are likely to have an even greater impact on the biotech industry, leading to the development of new and innovative therapies for a wide range of diseases.
Both antibody humanization and affinity maturation are critical steps in the development of therapeutic antibodies. However, traditional methods associated with these processes can be time-consuming and expensive. AI has revolutionized these processes by reducing the time and costs involved, as AI algorithms can analyze massive datasets of antibody molecules to identify the optimal amino acid sequences for humanization and affinity maturation. This information can be used to modify the antibody molecule, resulting in a more effective therapeutic agent.
Although the use of AI in antibody humanization and affinity maturation is still in its early stages, the potential benefits of this technology are clear. As AI algorithms become more sophisticated, they will be able to analyze even larger datasets and provide more accurate predictions, further reducing the time and costs involved in the antibody humanization and affinity maturation processes. AI is on course to become an essential tool in the development of therapeutic antibodies because of its ability to optimize antibody sequences and structures, identify novel targets, aid in repurposing of existing drugs and antibodies, and perform high throughput screening.







