Humanization of Antibodies
Enhancing Therapeutic Efficacy and Compatibility
Antibody humanization enhances the adaptability and efficacy of therapeutic antibodies derived from non-human sources, promoting safer and more effective applications in human medicine. This article explores the necessity of antibody humanization, reviews the evolutionary progression and development requirements of humanized antibodies, and examines various humanization strategies.
Evolution of Therapeutic Monoclonal Antibodies
The development of therapeutic antibodies has transitioned from mouse monoclonal antibodies to fully humanized forms, marking a significant advancement in biotechnology. Initially, antibodies were directly derived from mice and exhibited considerable immunogenicity in humans. The first generation of humanized antibodies, known as chimeric antibodies, involved replacing the constant regions of a mouse antibody with those of a human antibody, significantly lowering immunogenicity while retaining the mouse antibody’s high specificity. Subsequent generations, such as CDR-grafted antibodies, involve transplanting only the complementarity-determining regions (CDRs) of a mouse antibody onto a human framework, further reducing immunogenicity while aiming to maintain binding affinity. Surface-reshaped antibodies have been engineered by altering the surface amino acids of mouse antibodies to minimize epitope recognition by the human immune system, thus decreasing immunogenicity without compromising the antigenbinding site. Finally, fully human antibodies, generatedeither through phage display technology or from human B cells, are entirely derived from human sequences, providing the lowest immunogenicity risk and are optimal for repeated or long-term treatments.
Development Requirements of Humanized Antibodies
Humanized antibodies must adhere to stringent development standards to guarantee safetyand efficacy. Essential criteria include minimal immunogenicity, high binding affinity, and substantial in vivo stability. These antibodies should also be producible in large quantities under current Good Manufacturing Practices (cGMP) to maintain consistent quality and therapeutic effectiveness. Furthermore, extensive biocompatibility testing is required to verify that these antibodies do not elicit unintended biological responses.
Strategies of Antibody Humanization
Antibody humanization involves several sophisticatedtechniques, each designed to reduce the immunogenicity of non-human antibodies while preserving or enhancing their therapeutic efficacy. These methods are essential for developing antibodies that can be used safely and effectively in human medicine.
Complementarity-determining Region (CDR) Grafting
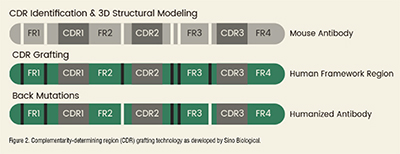
CDR grafting is the predominant method of humanization. This technique involves transferring the CDRs from a non-human antibody to a human antibody maintaining their integrity is essential for retaining the antibody’s specificity and affinity. The primarychallenge of CDR grafting is to incorporate theseregions into a human framework without altering theirstructure. Techniques such as advanced molecular modeling and X-ray crystallography are frequently used to ensure and validate the structural compatibility of the humanized CDRs within the human framework.
Specificity-determining Region (SDR) Grafting
SDR grafting refines the focus to the most critical parts within the CDRs—specifically, the amino acids that directly interact with the antigen. By modifying only these key areas, SDR grafting minimizes disruptions to the overall ntibody structure and function, thus preserving the original binding properties more effectively than broader CDR grafting approaches.
Resurfacing Approach
The resurfacing method alters the antibody’s external amino acids to reduce its immunogenicity without affecting the antigen-binding sites. This is achieved by replacing surface amino acids with variants more common in humans, thereby diminishing recognition by the human immune system. This approach isespecially beneficial when it is vital to preserve the integrity of CDRs to maintain binding affinity. Resurfacing can also be integrated with other grafting techniques to optimize outcomes.

Other Antibody Humanization Methods
Antibody library technologies utilize extensive collections of human antibodies to find sequences that serve as effective scaffolds for humanization. These libraries are generated either from B cells of immunized humans or by synthetic means such as phage display technology. Through meticulous screening, antibodies that exhibit desirable traits like high affinity, low immunogenicity, and specific effector functions are selected. This method is highly versatile and facilitates the rapid evaluation and optimization of numerous variants. Additionally, the use of transgenic mice engineered to produce human antibodies upon antigen exposure represents another innovative approach. These mice harbor uman immunoglobulin genes, producing antibodies that are inherently human and typically require minimal, if any, further humanization, offering a robust platform for generating fully human antibodies for therapeutic applications.
Humanization of Nanobodies (VHH)
Nanobodies, or VHH segments derived from camelid antibodies, are single-domain antibodies significantly smaller than conventional antibodies. Their compact size and unique structure enable access to cryptic epitopes inaccessible to conventional antibodies. Humanizing these nanobodies can substantially enhance their therapeutic potential by reducing immunogenicity and improving tissue penetration, making them particularly beneficial in targeted cancer therapies and diagnostic applications.
Applications of Humanized Antibodies
Humanized antibodies have transformed the treatment landscape for various diseases, especially in oncology and autoimmune disorders. These antibodies are engineered to target specific antigens, such as tumors or pathological immune cells, with high specificity, thereby minimizing side effects associated with traditional treatments. Additionally, humanized antibodies play a crucial role in the diagnostic process, where their specificity enables the detection of trace amounts of biomarkers.
Sino Biological provides alpaca nanobody and murine monoclonal antibody humanization services using complementarity-determining region (CDR) grafting technology and computer-aided molecular modeling, boasting a success rate of 100% and >95% sequence homology compared to human antibody frameworks.
Future Prospects
The future of antibody humanization looks promising, driven by continued advances in genetic engineering, molecular biology, and immunology. Emerging techniques such as next-generation sequencing are poised to further refine the precision and efficacy of humanized antibodies. The development of universal platforms for rapid antibody humanization is expected to reduce both the time and costs associated with antibody development, enabling quicker responses to emerging health challenges.










