The Future of Vaccine Development: How Antibody Profiling Addresses Key Challenges
Dr. Jonathan Blackburn, Chief Scientific Officer, Sengenics
Dr. Janique Peyper, Senior Bioinformatician and Systems Immunologist, Sengenics
Introduction
Vaccines are the most powerful tool in public health for controlling infectious diseases, preventing an estimated 154 million deaths worldwide over the past 50 years (1). Yet, despite their success, vaccine development faces significant challenges, from managing individual patient variability to addressing pathogen dynamics and ensuring long-term safety (Table 1). This article explores how antibody (Ab) profiling can address key obstacles in vaccine development, enabling researchers to design safer, more effective vaccines while streamlining the development process.
Identifying Effective Vaccine Candidates
The complexity of immune responses and the evolving nature of pathogens and tumors make it difficult to pinpoint the most effective vaccine targets. Ab profiling provides a powerful solution by detecting pre-existing or early-elicited protective Abs, such as those found in exposed but uninfected individuals. It can detect Abs that increase in concentration after exposure or vaccination in protected individuals, serving as important “correlates of protection.” Ab profiling can also directly identify immunogenic, disease-related antigens on the surface of target microbes and confirm the immunogenicity of selected vaccine targets, helping to lay the groundwork for more effective vaccine development.
Tackling Host Variability with Molecular Subtyping
Patient variability in immune responses—driven by differences in genetics, immune history, and HLA profiles—complicates the creation of universally effective vaccines. Ab profiling provides data that represent patient-specific immune responses, which can be associated with immune-influencing variables (e.g., HLA profile). Moreover, it facilitates immune-relevant population segmentation, helping to explain the variability in vaccine responses within a population. This approach in principle enables tailored vaccine design, ensuring vaccines are optimized not only for the general population but also for at-risk groups such as the elderly or immunocompromised.
Addressing Pathogen and Tumor Variability
Pathogens and tumors constantly evolve, posing another major hurdle for vaccine developers. By tracking Ab responses to emerging pathogens and neoplasms over time, researchers can dynamically monitor seroreactivity to ensure vaccines remain effective even in the face of antigen mutation and variability. Ab profiling can also identify which new pathogen or tumor antigens are immunogenic and which are not, based on cross-sectional analysis, as well as identifying individuals with broadly neutralizing antibodies capable of binding to multiple serotypes of the antigens.
Managing Prior Immunity and Tolerance
Pre-existing immune memory from previous infections or vaccines can lead to suboptimal responses, hindering progress in clinical trials for otherwise promising candidates. Ab profiling can identify prior seroreactivity to vaccine candidates or vectors, allowing for adjustments in vaccine design and selecting for patients who are more likely to respond favorably in clinical trials.
Enhancing and Monitoring Vaccine Safety and Efficacy
Ab profiling is an essential tool for early and ongoing evaluation of vaccine safety and long-term efficacy. In the preclinical stage, Ab profiling assists in detecting inappropriate cross-reactivity with host molecules or commensal organisms, helping researchers avoid potential autoimmune issues before clinical trials even begin.
During clinical trials, Ab profiling can provide critical insights regarding safety, dose, and route. For instance, profiling Abs across multiple biological compartments—such as oral, respiratory, gastrointestinal, and tissue microenvironments—allows researchers to evaluate how vaccination routes affect immune response, including reactive immunoglobulin isotypes and subclass combinations. This level of detail allows for the selection of the optimal vaccination route and ensures that the immune response is both robust and targeted. Additionally, it supports the creation of surrogate endpoints, identifying protective Abs produced following vaccination or booster administration, and revealing correlates of protection, thereby expediting clinical trials and reducing the overall time and cost of vaccine development.
Once a vaccine reaches the market, Ab profiling can track the long-term immune response, monitoring for vaccine-induced autoimmunity and assessing the continued efficacy of the vaccine against emerging pathogens.
Profiling Antibodies with Protein Microarrays
Protein microarrays offer a fast, cost-effective way to profile Abs against thousands of antigens simultaneously. However, to ensure accurate results, proteins must be correctly folded since 90% of Abs bind to conformational epitopes in vivo. This requires both proper protein folding and an array surface chemistry that preserves the structural integrity of these epitopes.
Sengenics sets itself apart with its proprietary KREX® technology, which guarantees that only full-length, correctly folded proteins are immobilized on a non-denaturing surface for Ab profiling (Figure 1). Misfolded proteins are washed away, preventing false positives and ensuring that only correctly folded, functional proteins are analyzed. This technology not only allows for the recognition of preserved in vivo epitopes by Abs, but also addresses a key challenge in vaccine research: generating stable, in vivo-relevant candidate protein antigens.
The platform offers outstanding performance, delivering high reproducibility (R² > 0.95), an extensive linear range exceeding 4 logs, and picomolar sensitivity, while requiring minimal sample volumes (<50 µL). Furthermore, Sengenics microarrays support the simultaneous analysis of two Ab isotypes, enabling a more comprehensive and nuanced understanding of immune responses—crucial for advancing vaccine development and immune monitoring.
This technology is available as ready-to-use panels or can be customized with specific protein antigens, supported through comprehensive services from study design to bioinformatics analysis.
Future Perspectives
The future of vaccine development will increasingly depend on Ab profiling through advanced tools, such as Sengenics protein microarrays, to address key challenges, including host variability, pathogen evolution, and safety concerns. By enabling earlier pre-selection as well as failure prediction, Ab profiling should help significantly reduce time spent on non-viable candidates. It can also enrich clinical trials by broadening patient inclusion, ensuring more diverse and representative study populations. Real-time response monitoring can help optimize vaccine efficacy and adjust strategies as needed, while accelerated development timelines and reduced costs improve overall efficiency. Ultimately, Ab profiling has the potential to contribute to regulatory approval by refining every stage of the vaccine development pipeline.
Table 1. How Ab Profiling Can Address Key Challenges in Vaccine Development
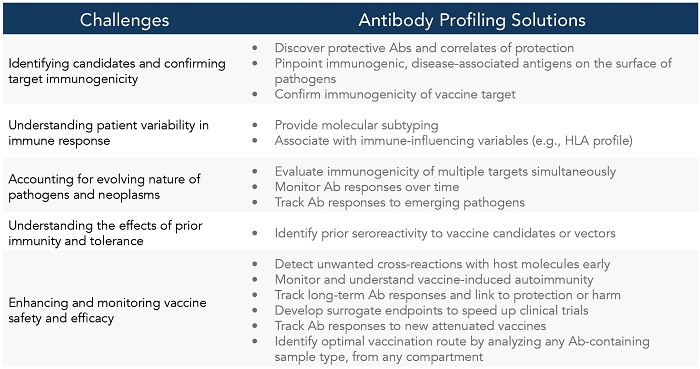
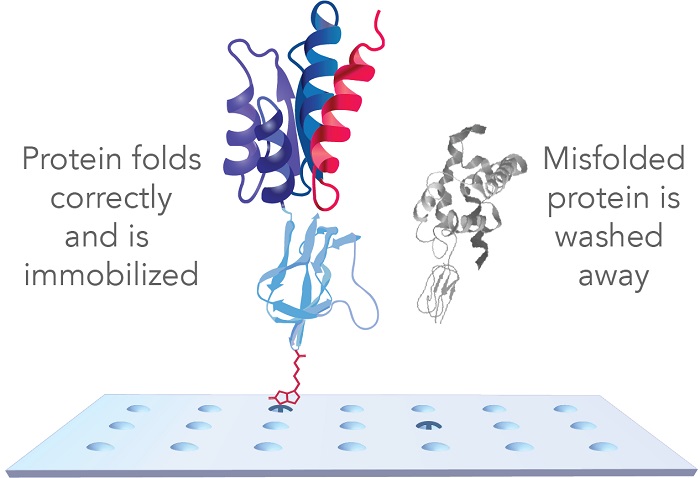
Figure 1. Sengenics protein microarrays feature functional, full-length, and correctly folded proteins for precision Ab profiling








