AI Drug Discovery IP Strategy: Navigating Patent Challenges
Prof. (Dr.) Amrish Chandra, Managing Director, Lexgin IP LLP
Artificial Intelligence is revolutionising drug discovery, offering unprecedented speed in target identification, compound optimisation, and clinical predictions. Yet, protecting these outputs through patents remains contentious. This article explores inventorship disputes, novelty challenges, disclosure hurdles, and global jurisdictional approaches while proposing strategies to safeguard AI-driven pharmaceutical innovations within evolving intellectual property frameworks.
Introduction
Drug discovery has historically been a costly, time-intensive endeavour. Traditional approaches relied on high-throughput screening, years of laboratory trials, and significant financial investment with high attrition rates. The advent of Artificial Intelligence (AI) promises to transform this paradigm. By integrating computational power with biomedical data, AI now allows researchers to identify promising molecules, model drug–target interactions, and predict patient responses far faster than human-led methods alone.
The pharmaceutical industry is embracing AI as a strategic asset. Companies such as Pfizer, Novartis, and Roche are investing heavily in AI platforms, while start-ups like BenevolentAI, Exscientia, and Insilico Medicine are making headlines with accelerated discoveries. However, while science advances rapidly, intellectual property (IP) systems remain firmly rooted in traditional concepts of human invention. Questions around ownership, inventorship, and reproducibility create barriers to patenting AI-driven discoveries. Unless addressed, these gaps risk undermining investment and slowing innovation.[1]
The Expanding Role of AI in Drug Delivery
AI applications span the full drug development pipeline:
- Target identification: AI analyses genomic and proteomic data to uncover disease mechanisms.
- Virtual screening: Billions of molecules can be computationally screened against biological targets in days.
- Lead optimisation: Predictive models refine compounds for efficacy, bioavailability, and safety.
- Drug repurposing: Algorithms discover new therapeutic uses for existing drugs, reducing costs and timelines.
- Clinical trials: AI predicts trial outcomes, identifies patient subgroups, and helps avoid costly failures.
Success stories underscore the impact. Exscientia advanced the world’s first AI-designed cancer drug (DSP-1181) into human trials in collaboration with Sumitomo Dainippon Pharma. DeepMind’s AlphaFold solved the protein-folding problem, long considered one of biology’s grand challenges, providing structural insights that accelerate drug target discovery. Insilico Medicine designed and pre-clinically tested a novel anti-fibrosis drug in less than 18 months—far faster than the typical multi-year process. Yet, these advances highlight the urgent need to align patent strategies with AI-driven processes.[2]
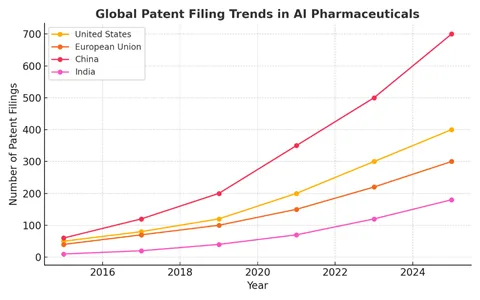
Figure 1: Global patent filing trends in AI pharmaceuticals [3]
Patentability Challenges
Inventorship dilemmas: Patent offices worldwide, including the USPTO, EPO, and UKIPO, have refused applications naming AI systems like “DABUS” as inventors. Their consistent position is that only humans can be inventors. For now, companies must attribute inventive contributions to individuals involved in designing, training, or interpreting AI outputs. This attribution requires careful documentation of human involvement in what may otherwise be machine-driven results.[4]
Novelty and Non-Obviousness: AI-driven outputs can blur the line between true invention and data-driven extrapolation. Patent examiners may view molecules generated by AI as “obvious” if they closely resemble known compounds. For example, if an AI predicts a derivative of an existing drug, the novelty requirement may fail despite therapeutic potential.
Disclosure and Reproducibility: Enablement is a cornerstone of patent law: an invention must be disclosed in sufficient detail for a skilled person to reproduce it. But AI models, particularly deep neural networks, often operate as opaque “black boxes.” Without explainability, applicants struggle to satisfy disclosure requirements. Patent examiners are increasingly asking not only what was discovered but also how it was derived.
Ethical and Commercial Considerations: Another emerging challenge is fairness. Many AI models are trained on open-access scientific data. Should companies gain exclusive patent rights over discoveries derived from public resources? This question resonates in both legal and ethical debates, influencing future reforms.
Global Jurisdictional Perspectives
United States: The USPTO requires human inventors. AI-assisted inventions are patentable if human researchers play a substantial role. Courts are beginning to explore disclosure standards in algorithm-heavy discoveries, and debates continue around how much algorithmic contribution qualifies as an “inventive step.”
European Union: The European Patent Office (EPO) applies a strict human inventorship rule. However, it protects AI-assisted inventions that provide a “technical contribution.” Abstract algorithms are excluded, but AI-enabled drug discoveries may qualify if tied to a clear therapeutic or industrial application.
China: China is actively encouraging AI innovation. While it also restricts inventorship to humans, patents for AI-enabled pharmaceutical discoveries are regularly granted if novelty and industrial utility are demonstrated. China’s proactive policies have spurred a surge in filings, making it a leader in AI-driven life sciences IP.
India: India excludes algorithms “per se” from patentability. However, applications where AI leads to tangible pharmaceutical outputs—such as novel compounds or therapeutic applications—may be accepted. Success hinges on careful claim drafting that emphasises the practical medical outcome rather than the underlying code.
Japan: Japan recognises the value of AI in drug discovery but, like most jurisdictions, restricts inventorship to humans. The Japanese Patent Office has issued guidance stressing the need for applicants to clearly identify human contributions in AI-driven processes.
South Korea: South Korea, a growing hub for biotech innovation, has taken a cautious approach. While patents for AI-enabled compounds are accepted, the nation has initiated consultations on whether to expand inventorship definitions in future.
Brazil: Brazil’s patent office has seen rising AI-related filings. Its stance mirrors other major offices: AI cannot be an inventor, but AI-assisted discoveries are patentable if human oversight and industrial application are demonstrated.[5]
Case Studies
BenevolentAI: Leveraged AI to propose a therapy for motor neuron disease. Human researchers validated the outputs, ensuring patent eligibility under current rules.
Insilico Medicine: Developed an AI-designed anti-fibrosis candidate and highlighted human oversight in data selection and validation, enabling robust patent claims.
Exscientia: Advanced the first AI-designed oncology drug into human trials. Its strategy combined patents for compounds with trade secrets for algorithms and datasets.
DeepMind’s AlphaFold: Solved protein-folding, producing a breakthrough tool. Its reliance on open scientific data has sparked debate on whether proprietary rights should extend to discoveries built on public resources.
Atomwise: Partners with pharmaceutical companies to screen massive chemical libraries. Licensing agreements clarify ownership of outputs, reducing disputes over IP.
Strategic Approaches to IP Protection
Emphasise Human Attribution: Organisations should ensure human roles are well documented throughout AI-assisted processes. Evidence of design, supervision, and decision-making strengthens inventorship claims.
Hybrid IP Models: Patents protect final outputs like molecules, while trade secrets safeguard proprietary algorithms and datasets. This layered approach reduces exposure to uncertainty in patent law.
Treat Data as Strategic IP: High-quality datasets are as valuable as the compounds they generate. Confidentiality agreements, licensing, and strong governance policies are essential to protect this resource.
Collaboration and Licensing Frameworks: AI start-ups and pharmaceutical giants often collaborate. Clearly defined licensing agreements should allocate rights over datasets, algorithms, and outcomes, preventing disputes.[6]
Policy Engagement: By engaging with regulators and industry groups, companies can shape evolving frameworks. Active participation ensures that industry realities are considered in future reforms.
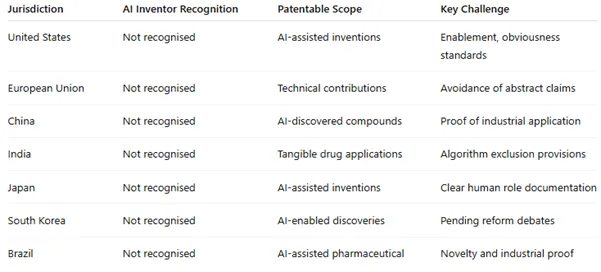
Figure 2: Comparative Jurisdictional Overview
Future Outlook
AI’s role in pharmaceutical research will only grow. This will force regulators to reconsider outdated frameworks. Potential developments include:
- Sui generis protections: New rights specifically designed for AI-generated outputs.
- Expanded inventorship definitions: Future reforms may consider recognising AI as a co-inventor, though this remains controversial.
- Global harmonisation: WIPO-led discussions are pushing for international alignment, reducing cross-border uncertainty.
- Ethical frameworks: Policymakers may introduce rules ensuring equitable benefit-sharing when AI is trained on public or open datasets.
- Commercial implications: Large pharmaceutical firms may adapt quickly, but smaller companies could struggle with the complexity and cost of dual patent–trade secret strategies.
These possibilities underscore the importance of a proactive strategy. Companies that anticipate reforms and engage in dialogue with regulators will be better placed to thrive in the shifting landscape.
Conclusion
AI is redefining the future of drug discovery, but intellectual property law is lagging behind. Questions of inventorship, novelty, and disclosure present formidable challenges. Until legal frameworks evolve, companies must adopt pragmatic IP strategies—emphasising human attribution, layering patent and trade secret protections, protecting datasets, and negotiating clear collaboration agreements.
The pharmaceutical industry must also participate in policy debates to ensure balanced reform. Only by bridging innovation and regulation can firms safeguard their AI-driven breakthroughs while ensuring fair and equitable progress in global healthcare.
References
- Y. Shimizu, M. Ohta, S. Ishida, K. Terayama, M. Osawa, T. Honma, and K. Ikeda, J Cheminform 15, 120 (2023).
- J. Wang, J Law Biosci 12, (2025).
- GreyB, "Artificial Intelligence (AI) Patent Landscape: Global Innovation," .
- A. Chaturvedula, S. Calad‐Thomson, C. Liu, M. Sale, N. Gattu, and N. Goyal, CPT Pharmacometrics Syst Pharmacol 8, 440 (2019).
- Amit Gangwal, Frontiers (Boulder) (n.d.).
- L. Schultz, Arizona Law Journal of Emerging Technologies 8, (2024).











