Applying CPCA: A Risk Based Framework for Nitrosamine Control in Pharmaceuticals
Subathra Ramamoorthy, Is a Senior Scientist at Veranova
As nitrosamine control shifts from reactive testing to a lifecycle, risk based obligation, the
Carcinogenic Potency Categorization Approach (CPCA) has emerged as a critical regulatory tool. This article shows how CPCA converts chemical structure into five potency categories to set AI limits and realistic analytical targets, strengthening nitrosamine risk management and ensuring patient safety.
1. Introduction: Why Nitrosamines Remain a Critical Quality Challenge
N-Nitrosamines are a structurally diverse class of potent mutagenic carcinogens formed when nitrosating species react with amines, often via α hydroxylation to DNA reactive intermediates. Even at trace levels, long term exposure in chronically administered medicines can translate into a measurable incremental lifetime cancer risk. The 2018 discovery of nitrosamines in valsartan and other sartan products was the inflection point that transformed nitrosamines from an occasional contamination issue into a global quality and supply chain vulnerability.
Since then, the focus has expanded beyond classical small nitrosamines such as NDMA and NDEA to complex nitrosamine drug substance related impurities (NDSRIs) that are specific to individual APIs, routes, and formulations. Nitrosamine control is now understood as a lifecycle obligation spanning development, manufacturing, packaging, and stability, not merely a specification and testing exercise. Against this backdrop, a nitrosamine specific, predictive framework became essential to support consistent, risk proportionate decisions.
2. Emergence of Nitrosamine Drug Substance Related Impurities (NDSRIs)
NDSRIs are N nitrosamines structurally related to the API or its key intermediates, typically formed when amine containing drug substance structures encounter nitrosating conditions somewhere along the synthetic or product lifecycle. In contrast to small dialkyl nitrosamines, NDSRIs often embody the full complexity of the drug’s scaffold, including heterocycles, multiple functional groups, and stereochemical features. Their formation can depend on subtle process variables such as residual nitrite, amine salt equilibria, excipient nitrite levels, and interactions with packaging or storage conditions.
Most NDSRIs lack robust compound specific carcinogenicity data, and in many cases even high quality structural analog data are limited. Nevertheless, regulators and companies must still set acceptable intake (AI) limits and implement control strategies that protect patients while preserving supply. The classical ICH M7 toolkit—read across, QSAR, and TTC—was not built to handle nitrosamines’ extreme potency distribution and nitrosamine specific mechanistic features. This created a need for a dedicated framework that could extend the available data to structurally similar, data poor nitrosamines in a transparent and reproducible way.
3. Why CPCA Was Introduced: Addressing Data Gaps in Nitrosamine Risk Assessment
The Carcinogenic Potency Categorization Approach (CPCA) was developed to answer a simple but critical question: how can regulators and manufacturers recommend acceptable intake (AI) limits for nitrosamines when the vast majority lack direct carcinogenicity data? CPCA was co developed by FDA and the Nitrosamine International Technical Working Group (NITWG), a consortium of international drug regulatory authorities, to provide a common, structure based framework for predicting nitrosamine potency and recommending AI limits.
Rather than treating all nitrosamines as equally potent or relying solely on generic TTC values, CPCA leverages a curated database of nitrosamine carcinogenicity studies and well established structure–activity relationships centered on α hydroxylation mediated activation. It is implemented as a rule based SAR framework: defined structural features around the N nitroso group are evaluated and used to assign each nitrosamine to one of five potency categories (PC1–PC5), each linked to a default AI range. The goal is to provide conservative, scientifically grounded starting AI recommendations that are more differentiated than a single TTC value yet still practical to apply across many nitrosamines, filling the gap where compound-specific data are absent.
It has since been adopted or explicitly referenced in nitrosamine guidance by multiple global regulators, including EMA, Health Canada, and WHO, and is increasingly used as a global reference approach for setting AI limits.
4. What CPCA Is — and What It Is Not
Conceptually, CPCA is a categorical binning framework, not a bespoke AI calculator for every nitrosamine. It is designed for N nitrosamines that operate via the canonical α hydroxylation activation mechanism, such as N nitrosamines of secondary and certain tertiary amines. For each nitrosamine within this domain, structural features are mapped to a “potency score,” which then determines assignment to PC1–PC5, with lower category numbers corresponding to higher predicted carcinogenic potency.
Importantly, CPCA does not override robust compound specific animal carcinogenicity data or AI limits already established in regulatory guidance or product specific assessments. Where such data exist, they should form the primary basis of AI derivation. CPCA is intended only for N nitrosamines that undergo α carbon hydroxylation and should not be applied to nitrosamides or other nitroso chemotypes that follow different bioactivation mechanisms.
5. The CPCA Binning Framework and Structural Feature Considerations
At the heart of CPCA is a structured evaluation of features that influence nitrosamine carcinogenic potency. The framework can be viewed as a guided decision tree that considers four key elements.
Scope and mechanism
- Is the impurity an N nitrosamine likely to be activated via CYP mediated α hydroxylation, leading to a diazonium ion or related electrophile capable of alkylating DNA?
- Nitrosamides and other nitroso classes that do not follow this mechanism fall out of scope and should not be categorized by CPCA.
α-Hydrogen features
- Presence, number, and substitution pattern of α hydrogens on the carbon(s) adjacent to the nitrosated nitrogen.
- α Hydrogens at benzylic, allylic, or otherwise activated positions are associated with higher carcinogenic potency due to facilitated α hydroxylation and more stable cationic intermediates.
Activating features
- Electron donating groups or conjugated systems near the N nitroso group that increase electron density and promote metabolic activation.
- Structural motifs historically associated with potent nitrosamines, such as certain aromatic or heteroaromatic substituents in conjugation with the α carbon.
Deactivating features
- Strong electron withdrawing substituents that reduce electron density and α hydroxylation propensity.
- Steric hindrance around the α positions that limits enzyme access.
- Absence of α hydrogens (for example, quaternary centers), which may drastically reduce or eliminate the canonical activation pathway.
These features are combined into a potency score using predefined rules. This score is then mapped to one of five potency categories, each associated with a default AI range, as summarized below.
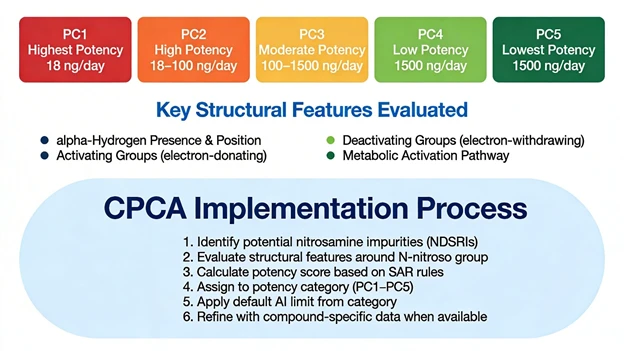
The numeric boundaries reflect statistical analysis of nitrosamine carcinogenicity datasets and are anchored to conservative human cancer risk benchmarks. For NDSRIs, small structural modifications—such as introducing or removing an activated α carbon or changing electron donating or electron withdrawing substituents—can shift an impurity across categories, with direct consequences for its AI. This sensitivity underscores the importance of high confidence structural elucidation and careful mechanistic interpretation.
6. Applying CPCA in Practice: From AI Limits to Analytical Targets
For industry practitioners, CPCA is most useful when embedded in a structured risk management workflow rather than treated as a one off classification step. A typical sequence begins with structural definition and confirmation: plausible nitrosamine structures are identified from the synthetic route, reagents, excipients, packaging, and degradation pathways, and key candidates are confirmed using MS, NMR, or other orthogonal techniques where feasible.
Next, CPCA rules are applied to each confirmed or reasonably suspected nitrosamine to assign a potency category (PC1–PC5) and select the corresponding default AI as a conservative starting point, with assumptions and borderline judgments clearly documented. CPCA outputs are then combined with clinical exposure parameters (dose, regimen, population) to prioritize products and sites by nitrosamine risk, focusing resources first on high exposure, high potency (PC1–PC2) scenarios.
The final step is translation into analytical targets. Default AIs (ng/day) are converted into required product concentrations by considering maximum daily intake and relevant worst case scenarios, and analytical target profiles and LOQs are set to ensure that validated methods can realistically achieve the necessary sensitivity across all relevant matrices.
These linkages—CPCA category → AI → product concentration → LOQ—should be traceably documented in risk assessments, validation reports, and technical agreements so that CPCA actively drives analytical and process decisions rather than remaining an isolated theoretical exercise.
7. Common Pitfalls in CPCA Implementation
Experience has revealed several recurring pitfalls that can undermine CPCA’s intent. One is optimistic “bin shopping”: assigning a less conservative potency category by over weighting deactivating features or under interpreting activating motifs, particularly when structures are incompletely characterized, which can yield under protective AIs and draw regulatory scrutiny when patient exposure is non trivial. A second is neglecting metabolic plausibility—applying CPCA to nitrosamines that may not follow the α hydroxylation pathway or ignoring alternative activation routes—leading to questionable category assignments.
A third pitfall is disconnecting CPCA from analytical reality by setting very low CPCA based AIs without confirming that validated methods can reach the corresponding LOQs in all relevant matrices, creating a gap between nominal and actual control. A fourth is failing to plan for refinement, treating CPCA as the final answer for high impact products (for example, large patient populations, limited alternatives, long term use) and remaining locked into overly conservative limits longer than necessary.
Preventing these issues requires cross disciplinary governance. Chemists, toxicologists, analytical scientists, and regulatory experts should jointly review CPCA categorizations, challenge optimistic assumptions, and ensure that expectations around CPCA application, conservative defaulting, and refinement are clearly defined, including in sponsor–CDMO quality and technical agreements.
8. When CPCA Must Be Refined or Replaced
There are clear situations in which CPCA based AIs should be refined or replaced by more specific assessments. High or unavoidable exposure—when worst case or actual patient exposure approaches or exceeds a CPCA based AI—may warrant refinement to avoid either over conservatism that risks supply or under protection of patients. Structural and mechanistic uncertainty, such as multiple plausible NDSRIs, uncertain nitrosation sites, or potential non canonical mechanisms, can similarly limit confidence in a simple CPCA bin. Regulatory feedback or new carcinogenicity or mutagenicity data that challenge existing assignments are also triggers for reassessment.
Refinement options include read across using robust comparisons to structurally similar nitrosamines with high quality carcinogenicity data; enhanced Ames and related in vitro assays to distinguish high from lower potency members; and targeted in vivo studies in high impact cases. CPCA should therefore be viewed as part of a dynamic evidence hierarchy: as new data emerge, nitrosamines can move between potency categories and their AIs recalibrated, supported by a transparent narrative from initial binning through refinement for both internal stakeholders and regulators.
9. CPCA in a Lifecycle Nitrosamine Control Strategy
In a mature quality system, CPCA is embedded within a broader lifecycle nitrosamine control strategy rather than used as a one time exercise. During development, CPCA can inform route and formulation design by highlighting high risk amine–nitrite combinations or structural motifs best avoided early, and it supports triage of potential NDSRIs, guiding how deeply they should be characterized and followed in stability studies.
In commercial manufacturing, CPCA outputs should feed into ongoing risk reviews, process changes, site transfers, and supplier qualification, with reassessment of plausible NDSRIs and their categories whenever materials, processes, or suppliers change. Within a Quality Risk Management framework, CPCA becomes one of several inputs—alongside process knowledge, analytical capability, and clinical context—that collectively shape the nitrosamine control strategy and provide a common technical language between sponsors and CDMOs for roles, timelines, and refinement expectations.
10. Conclusion: CPCA as a Marker of Risk Based Quality Maturity
CPCA represents a significant evolution in how the industry manages nitrosamine impurities under conditions of structural complexity and incomplete data. By distilling a substantial carcinogenicity evidence base and mechanistic understanding into a five bin potency framework, it enables consistent, transparent, and conservative AI recommendations across a wide range of nitrosamines.
Its value, however, depends on how rigorously it is applied. Organizations that use CPCA as a static lookup tool, disconnected from structural rigor, metabolic plausibility, and analytical capability, risk both under and over control. In contrast, companies that integrate CPCA into lifecycle nitrosamine control—coupling it with robust structural elucidation, realistic method development, and timely refinement—demonstrate a higher level of risk based quality maturity and an ability to engage regulators with a defensible, data driven narrative on nitrosamine risk.
References:
- https://www.fda.gov/regulatory-information/search-fda-guidance-documents/control-nitrosamine-impurities-human-drug
- https://www.ema.europa.eu/en/documents/referral/nitrosamines-emea-h-a53-1490-questions-answers-marketing-authorisation-holders-applicants-chmp-opinion-article-53-regulation-ec-no-726-2004-referral-nitrosamine-impurities-human-medicinal-products_en.pdf
- https://extranet.who.int/prequal/news/update-n-nitrosamine-impurities
- https://database.ich.org/sites/default/files/ICH_M7-R2_Guideline_Step4_2023_0323.pdf
- https://pubmed.ncbi.nlm.nih.gov/39662665/; doi: 10.1016/j.yrtph.2024.105762. Epub 2024 Dec 9.
- https://pubmed.ncbi.nlm.nih.gov/38754805/; https://doi.org/10.1016/j.yrtph.2024.105640










