Artificial Intelligence in Clinical Trials
Reva Rajendra Joshi, Masters Student at the MIT World Peace University School of Pharmacy
Ashwin Kuchekar, Associate Professor and the Head of Career Services and Placement at the School of Pharmacy
Artificial intelligence is a computer program that extracts concepts and relationships from data and learns on its own from data patterns. A clinical trial is a time-consuming, costly process with a very low success rate. Artificial intelligence will overcome challenges associated with clinical trials and will reshape the pharmaceutical industry’s future. Currently, many pharmaceutical industries started to invest in AI to improve pharmaceutical research.
Artificial intelligence is a field of engineering and science that focus on making intelligent machines. Artificial intelligence is a top technology that is reshaping the pharmaceutical industry's future. For ages, the pharmaceutical industry has been developing cures and treatments. Traditionally, medication design and manufacture took many years, extensive clinical studies, and sky-high prices.This has been changing with the advancement of 21st-century technology. We will see different drug designs, manufacturing, and clinical trials in the future.
Natural Language Processing (NLP), Machine learning (ML), Optical Character Recognition (OCR), and Deep Learning (DL), are examples of AI approaches. Clinical development in the traditional way is a time-consuming process with just a 10% success rate. Artificial intelligence (AI) can reduce the cost and burden of clinical development while improving the clinical phases of trials. Now many big pharmaceutical companies began to invest in AI to gear up pharmaceutical research.1
Problems with the traditional clinical trial process-
For the drug development process average 10-15 years are required in which 5-6 years are required for R&D and another 5-7 years are required for clinical trials and billions of monies (around USD 1.5-2 billions) spend on clinical trial process and success rate to become regulatory approved drug is just 10%.Major cost drive for a clinical trial process is patient recruitment about 32% of money spend on it.2
It is difficult to mine numerous data sets for clinical trials and keeps data of each patient involved in trial process so, artificial intelligence is helpful to overcome all these problems with the clinical trial process.
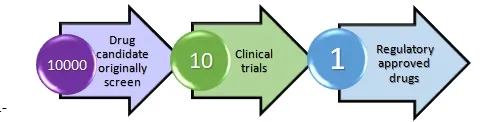
Figure1-Traditional clinical trial process
In future AI adoption will become vital need in following areas-
1. Design of clinical trial- AI has given rise to new tools that can collect, organise, and interpret the rising data generated by clinical studies, even from unsuccessful studies, and get meaningful data patterns which help to design.
2.Enrichment and recruitment of patients-Through the collection, analysis, and interpretation of many data sources, like medical imaging, 'omics' data and electronic health records (EHRs) Artificial intelligence digital transformation can enhance the patient selection process and improve clinical trial efficacy.Identifying appropriate individuals can speed up and enhance the efficiency of a clinical trial process.
3. Investigator and site selection-AI assist biopharma businesses in identifying target sites, priority candidates, qualified investigators and collecting and collating information to convince regulators that trial procedure follows GCP guidelines.
4. Monitoring of patient, drug adherence and retention-By automating data collecting, data exchange across systems, and digitising common clinical examinations, AI algorithms can aid in the monitoring and management of patients. Real time insights, regularly patient monitoring on the safety and effectiveness of therapy, as well as the danger of dropouts, may be enabled by AI algorithms combined with wearable technology, resulting in increased engagement and retention.
5.Strategic relationships and outsourcing to get necessary Artificial Intelligent skills and talent-Contract Research Organizations (CRO) that have made investments in data science capabilities are increasingly seen as key partners, offering access to not just specialised knowledge but also large number of people who wish to taking part in trials. Biopharmaceutical businesses caught the interest of tech giants. In addition to collaborating or contracting with biopharma, digital technology firms are increasingly operating in the clinical trials area. These collaborations integrate the fundamental strength in digital science of Tech giants and start-ups with the skills and knowledge in the medical science of biopharma.3
Future clinical trials-
Future AI will combine with improved developments in personalized medicine and computer simulation to create in silico trials.4 Virtual trials take advantage of the potential of modern digitized technologies to reduce the cost, time and difficulties which patients face. Virtual trials allow for more representative groups to be enrolled in real time and in their natural surroundings, as well as remote monitoring of these patients. Virtual trials might contribute for up to half of all studies, boosting patient retention and shortening clinical development timelines.5
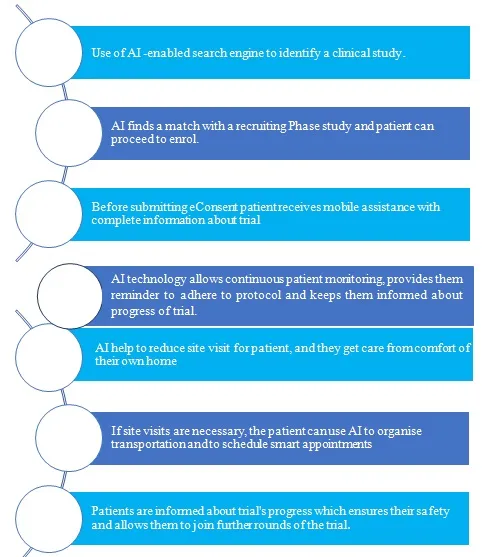
Figure 2- Future clinical trial process with the help of AI
AI is doubtless future of pharma industry. AI will save human effort, time and money. Now many big pharmaceutical companies began to investing in AI. Although artificial intelligence is still to be widely adopted and used in clinical studies.AI has the power to revolutionize clinical research. Application of AI result in quicker, safer, and less expensive clinical trial.AI will overcome problems associated with clinical trials.
References-
1. Taylor K, Properzi F, Cruz MJIntelligent clinical trials: transforming through AI-enabled engagement.2020
2. Calaprice-Whitty D, Galil K, Salloum W, Zariv A, Jimenez B. Improving clinical trial participant prescreening with artificial intelligence (AI): a comparison of the results of AI-assisted vs standard methods in 3 oncology trials.2020.
3. Harrer S, Shah P, Antony B, Hu J. Artificial intelligence for clinical trial design. Trends in pharmacological sciences. 2019;40(8):577-91
4. Beck JT, Vinegra M, Dankwa-Mullan I, Torres A, Simmons CC, Holtzen H, Urman A, Cognitive technology addressing optimal cancer clinical trial matching and protocol feasibility in a community cancer practice.2017.
5. Lee Y, Dinakarpandian D, Katakam N, Owens D. MindTrial: An intelligent system for clinical trials. InAMIA Annual Symposium Proceedings 2010.











