Innovations in Antibody-Drug Conjugates: A Deep Dive into Pharma's Future
Kate Williamson, Editorial Team, Pharma Focus America
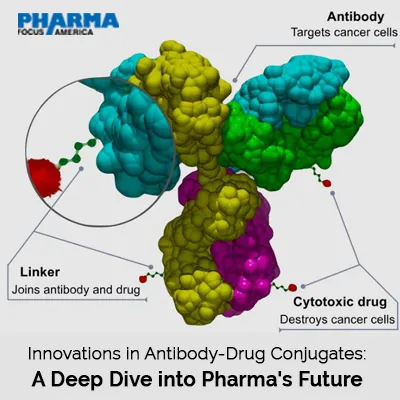
Antibody-Drug Conjugates (ADCs) for cancer therapy, are truly a hybrid of both monoclonal antibodies as well as cytotoxic drugs. In this article, we shall focus on the future developments of the ADC technology, as well as on the newest breakthroughs, their efficacy and limitations, providing readers with the clear vision of the main strategies of the upcoming pharma evolution.
Introduction: The Promise of Precision in Oncology
In the era of fast changing treatment paradigms of oncology, Antibody-Drug Conjugates (ADCs) are the novel type of targeting therapies. Through the incorporation of the targeted ability of monoclonal antibodies with the effectiveness of highly toxic payloads, ADCs are potential to transform current cancer therapies. This innovation solves one of the main problems of traditional chemotherapy: non-selectivity, as toxic drugs affect only cancerous cells without harming healthy cells. As the pharma industry starts looking into ADCs, a new trend of innovation is making the way these products work more efficient, safer, and diverse.
The Evolution of ADCs: From Concept to Clinical Success
The idea, with which everyone today familiar is called ADCs, launched its either many years ago with the thought of using therapeutic agents to target cancer cells like a “magic bullet”. Early trials but were associated with problems which include instabilities, unwanted side effects and lower effectiveness. The increasing interest in targeted therapy began with the FDA approval of Adcetris (brentuximab vedotin) in August 2011 and Kadcyla (ado-trastuzumab emtansine) in August of 2013. These successes created a lot of interest which resulted in the creation of more complex ADCs. Today, the field appears with over 14 approved ADCs and a rich portfolio of candidates spanning various therapeutic classes.
Innovations in ADC Design: Building the Next Generation
Enhanced Antibody Engineering
Today’s ADCs, as compared to their first-generation analogs, benefit from enhanced target selectivity and enhanced binding kinetics. Scientists prefer developing humanized or fully human antibody to avoid immunogenic reaction and consequently produce better therapeutic benefits. To improve the targeting of the heterogeneous tumors, bispecific antibodies, which can bind to two different antigens, are also under investigation.
Novel Linker Technologies
The spacer arms which join the antibody to the cytotoxic payload are points of concern in ADC stability and therapeutic outcomes.
New linker design has continued to evolve, and this has enhanced accrute flow of drug release with cleavable and non-cleavable linkers. Cleavable linkers discharge the payload at once when tumor-specific stimuli are triggered such as low pH values or enzymes while non-cleavable linkers allow stability and retention within the tumor without releasing the payload in circulation.
Payload Advancements
The target moiety, the payload, is often a cytotoxic drug that is incorporated into ADCs. Advancements have shifted to develop more potent payload with scheme of action different from the previous ones with addition of DNA alkylators, tubulin inhibitors and topoisomerase inhibitors. Other combinations include dual payload strategies whereby ADCs engage cancer cells in more than one way possible.
Site-Specific Conjugation
Conventional approaches of linking the payloads to the antibodies lead to heterogeneous ADC, which is a concern to their quality and the desired therapeutic ratio. Unlike PEGylation where polymer chain length constitutes the conjugation site, site-specific conjugation methods include cysteine-engineered antibodies and enzymatic conjugation to attach payloads more efficiently hence producing more cohesive ADCs.
Expanding the Therapeutic Horizons
Beyond Oncology
Though ADCs continue to be developed mainly for oncology applications, they are now being tested even in non-oncology areas such as infectious disease and autoimmune disease. For instance, researchers are using ADCs for antibacterial effect for pathogens causing bacterial infections, or to manage inflammation in rheumatoid arthritis.
Combination Therapies
Integration of ADCs with other therapies is a new approach to improve outcomes: ADCs with other chemotherapy agents or with inhibitors of immune check points. This interaction can synchronize the response of immune system to create a better attack on tumors.
Personalized Medicine
The discovery of biomarkers in combination with diagnosis instruments is creating a new trend in ADC treatments. This can be attributed to early recognition of patients with unique tumor markers to avail the precision medicine of bigger effectiveness and smallest side effects.
Overcoming Challenges in ADC Development
Unfortunately, ADCs suffer from a number of problems that offer no easy solution. Nevertheless, there are some challenges that are worth acknowledging in a more detailed manner: the key barrier today is the emergence of resistance when the cancer cells start to learn how to avoid ADC mechanisms. Scientists are solving it through combination therapies and next generation payloads.
Others are the issues of the steep toxicity profile, where the differences between therapeutic and toxic effects are quite small.
Emerging linker technologies and payload structures are in fact vital in extending this window. More so, the creation of ADCs requires the employing of multiple processes which might have impacts on both the scale and cost of production. To address these barriers, there has been ongoing growth in technologies associated with bioprocessing.
Regulatory Landscape and Market Trends
The dynamics of ADC regulation correspond to the dynamics of ADC development. USFDA and EMA have laid down some parameters of standard practices in order to ease the process of approval of ADCs based on certain principles regarding safety, efficacy and quality of manufacturing. Global addressable ADC market is also expected to rise dramatically over the years due to a broadening product pipeline, higher incidences of cancer, and higher funding in the biopharmaceuticals industry.
Most major stakeholders in the ADC space, including large pharma companies and biotechs, are partnering in order to bring ADCs along more quickly. A prime example of industry collaboration is the partnership between AstraZeneca and Daiichi Sankyo that has co-birthed a blockbuster ADC named Enhertu.
The Future of ADCs: A Vision for Pharma’s Frontier
During the recent years, AI and ML have become integrated in ADC field and they pose the capability to transform drug discovery and development. These technologies can enhance target recognition, rationalize designing procedures, and foretell clinical repercussions apace with earlier never before seen precision.
Furthermore, advancements in delivering systems, including ADCs that are based on nanoparticles, are expected to improve the delivery effectiveness and lower toxicity. Cancer researchers are also examining the application of ADCs with new treatment approaches such as cell and gene therapy and may expand the horizons for such therapies.
Conclusion: Embracing the Potential of ADCs
Antibody-Drug Conjugates are one of the most revealing and innovative forms of the new molecular revolution within the pharmaceutical industry that combines biology, chemistry, and technology systems. With the capability of targeting people’s medical needs that are yet unaddressed and opening new therapeutic options, ADCs can actually transform the prospects of future medicine. With this kind of developments increasing the pace in this field, the journey of ADCs shows the effectiveness of science and science alone and cooperation for a better tomorrow.










