One Health and Antimicrobial Resistance
Ramendra Pati Pandey, Assistant Professor, Department of Microbiology, SRM University, Rajiv Gandhi Education City, India
Gajala Deethamvali Ghouse Peer, Centre for Drug Design Discovery and Development (C4D), SRM University, Delhi-NCR, Rajiv Gandhi Education City, Haryana, India
Carlos R Prudencio, Center of Immunology, Institute Adolfo Lutz, São Paulo, Brazil
Antonio Charlys da Costa, Institute of Tropical Medicine, University of São Paulo, Brazil
Elcio Leal, Laboratory of Viral Diversity, Institute of Biological Sciences, Federal University of Pará, Brazil
Chung-Ming Chang, Master & Ph.D. program in Biotechnology Industry, Chang Gung University,Taiwan
It is for the most part recognized that we share many elements with other creature species, which are surely pertinent when the ideas of health and disease are talked about. The term 'One Health' is utilized in various settings and by individuals with changing foundations. Universally, an expected half of all antimicrobials serve veterinary purposes. Microscopic organisms that definitely create anti-microbial obstruction in creatures involve food-borne microbes, artful microorganisms and commensally microscopic organisms. The collection of safe microscopic organisms by the utilization of antibiotics in farming and veterinary medication and the spread of such microscopic organisms through farming and direct tainting are archived. Antimicrobial resistance keeps on expanding while the advancement for new antibiotics improvement is evaporating; after just eighty years of antibiotics use, bacterial diseases that used to be handily treated are becoming untreatable.. The fruitful results of numerous surgeries and immunosuppressive medicines rely upon anti-microbial prophylaxis and the capacity to treat infective entanglements. Anti-microbial resistance, subsequently, represents a genuine danger to quite a bit of medical services as far as we might be concerned. Anti-microbial resistance corresponds with antibiotic use, so that improved antimicrobial stewardship, with better counteraction and analysis of disease, can assist with rationing at present accessible antimicrobial specialists.
1. Introduction
Antibiotic resistance is anything but another wonder. Public observation information and autonomous examinations show that drug-safe, infection causing microscopic organisms have increased and spread at disturbing rates in late many years. An assorted scope of patients is influenced. The Institute of Medicine (IOM), Centers for Disease Control and Prevention (CDC), National Institutes of Health (NIH), and Food and Drug Administration (FDA) caution that drug-safe microscopic organisms are a genuine general health danger, particularly thinking about that there are a couple of novel medications ready to fight against these resistance microbes1. Studies connecting animals and human profiles of resistance have been founded overwhelmingly on aberrant affiliations4.
The One Health approach intends to achieve ideal health for individuals, creatures, and the climate2. Antibiotic resistance (ABR) is perceived as a One Health challenge in light of the quick rise and dispersal of safe microscopic organisms and qualities among people, creatures, and the climate at a worldwide scale3. Antimicrobials have empowered the headway of numerous spaces of clinical practice. Numerous methods of resistance are regularly present in a solitary strain of microscopic organisms, and this may likewise be joined with an increment in virulence, the two of which are prompting a critical expansion in mortality and morbidity in patients. Against this foundation, unquestionably the quantity of new anti-toxins authorized has declined particularly for Gram-negative multidrug-safe microbes. The purposes behind this disappointment are market issues, large pharma changes, administrative constraints, hardships in discovering drugable targets, and, ultimately, appropriate mixtures deserving of full turn of events1. Antibiotic revelation, methods of activity, and mechanism of resistance have been useful exploration subjects in scholarly communities5 and, as of not long ago, in the drug business. As regular items, they furnish testing scholarly exercises and shocks regarding their synthetic nature, biosynthetic pathways, advancement, and biochemical method of activity6, 7. The complete combination of such normal items in the research facility is troublesome, since these little molecules are regularly amazingly unpredictable in usefulness and chirality8.
2. Antibiotic resistance
Antibiotics have altered medication in many regards, and incalculable lives have been saved; their revelation was a turning point in mankind's set of experiences. Deplorably, the utilization of these marvel drugs has been joined by the quick appearance of safe strains. Clinical intellectuals are currently cautioning of a re-visitation of the pre-antibiotic time; a new data set records the presence of something else than 20,000 potential resistance qualities of almost 400 various sorts anticipated in the principle from accessible bacterial genome groupings9. Luckily, the number existing as practical resistance determinants in microorganisms is a lot more modest.
Table 1: Commonly used antibiotics and their Modes of action and resistance mechanisms
| Antibiotic class | Target | Mode(s) of resistance | Examples |
| β-Lactams | Peptidoglycan biosynthesis | Hydrolysis, efflux, altered target | Penicillins (ampicillin), cephalosporins (cephamycin), penems (meropenem), monobactams (aztreonam) |
| Aminoglycosides | Translation | Phosphorylation, acetylation, nucleotidylation, efflux, altered target | Gentamicin, streptomycin, spectinomycin |
| Glycopeptides | Peptidoglycan biosynthesis | Reprogramming peptidoglycan biosynthesis | Vancomycin, teicoplanin |
| Tetracyclines | Translation | Monooxygenation, efflux, altered target | Minocycline, tigecycline |
| Quinolones | DNA replication | Acetylation, efflux, altered target | Ciprofloxacin |
| Sulfonamides | C1 metabolism | Efflux, altered target | Sulfamethoxazole |
| Lipopeptides | Cell membrane | Altered target | Daptomycin |
Microbes can be inherently impervious to specific antibiotics yet can likewise get resistance to antibiotics by means of changes in chromosomal qualities and by even quality move. The inborn resistance of bacterial animal categories to a specific antibiotic is the capacity to oppose the activity of that antibiotic because of intrinsic underlying or useful attributes. The model identifies with the lipopeptide daptomycin (first supported for clinical use in 2003), which is dynamic against Gram-positive microscopic organisms however isn't compelling against Gram-negative microbes. This is because of an inherent contrast in the structure of the cytoplasmic layer; Gram-negative microscopic organisms have a lower extent of anionic phospholipids in the cytoplasmic layer than do Gram-positive microscopic organisms, which diminishes the effectiveness of the Ca2+-interceded addition of daptomycin into the cytoplasmic layer that is needed for its antibacterial activity11. The inherent resistance of some Gram-negative microscopic organisms to many mixtures are because of a failure of these specialists to cross the external layer: for instance, the glycopeptide antibiotic vancomycin hinders peptidoglycan crosslinking by restricting to target D-Ala-D-Ala peptides yet is just regularly viable in Gram-positive microorganisms as, in Gram-negative organic entities, it can't cross the external film and access these peptides in the periplasm12. The antibiotics with their mode of action and their resistance mechanism has presented in Table 1.
There are different mechanism where the microbes undergoes to get resistance to the antibiotics like by Preventing of access to target of the antibiotic by Reducing permeability where the microbes forms the permeability barrier from the antibiotics on their outer membrane13, 14. By increasing efflux, Bacterial efflux pumps out effectively transport numerous antibiotics from their cell and is significant supporters of the inherent resistance of Gram-negative microscopic organisms to a large number of the drugs that can be utilized to treat Gram-positive bacterial diseases. When over expressed, efflux pump can likewise present undeniable degrees of resistance to already clinically valuable antibiotics. Some efflux siphons have tight substrate, yet many vehicles a wide scope of fundamentally divergent substrates and are called as multidrug resistance (MDR) efflux pumps. By mutation the microbes undergoes the change in the antibiotic target site. By this mutation antibiotic losses its function and thus the microbe gets resistance to this antibiotic by changing the target site16. There are few microbes which directly modifies antibiotics preventing them from entering into the cell or altering their targets. By this they can destroy or modify antibiotics so that they can resist their action. These can be done by hydrolysis or by the chemical action17.
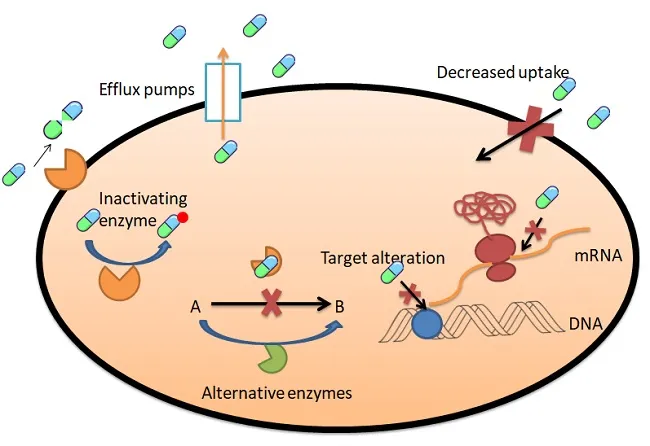
Figure 1: Different Molecular mechanism of antibiotic resistance
3. Antibiotic resistance in Animals
The advancement of antibiotic resistance in microorganisms has been ascribed to the utilization of antimicrobials in human medication. The commitments of veterinary medication and farming to anti-toxin resistance are as yet being examined. The veterinary utilization of antibiotics remembers the utilization for pets, livestock, and creatures brought up in hydroponics. For livestock, anti-infection agents are utilized in treatment and prophylaxis, and to build development and feed efficiencies. The fundamental irresistible illnesses treated are enteric and pneumonic contaminations, skin and organ abscesses, and mastitis. Anti-infection agents utilized in both veterinary and human medication are: penicillins, cephalosporins, nitrofurans, nitroimidazoles, sulfonamides, trimethoprim, polymyxins, and quinolones22. Of the more than 1 million tons of anti-infection agents delivered into the biosphere during the most recent 50 years23, generally half are assessed to stream into the veterinary and farming channels24. The use of antibiotics at subtherapeutic levels for expanded development and feed efficiencies in livestock (pigs, dairy cattle, turkeys, and chicken), a coordinated piece of present day agribusiness around the world, is exceptionally questionable25.
The molecular examination of antibiotic resistance qualities, plasmids, and transposons has exhibited that indistinguishable components are found in creatures and people. The marvel applies to pathogenic (for example foodborne), crafty and commensal microbes: the utilization of antibiotics in veterinary medication, like its utilization in farming and hydroponics, chooses for safe microscopic organisms. These microbes are delivered into the climate, where they can be effectively illustrated, in creature dung. Explicit food things, water, and direct contact can spread these microbes from creature microflora to human microflora. End of resistance determinants from these microflorae is slow, especially in case there is no repository of defenseless microbes accessible to recolonize the host creature. One inquiry to be addressed is whether commensal microbes from food can move their resistance qualities to the native human microflora during intestinal travel. Judicious utilization of antibiotics is suggested and ought to be sought after until we have precise measurements on the quantitative commitment of veterinary medication and farming to anti-toxin resistance26.
4. The role of One Health
Human populace increment, industrialization, and international issues speed up worldwide changes making critical harm biodiversity, broad disintegration of environments, and extensive transient development of both humankind and species overall. These fast ecological changes are connected to the rise and reappearance of irresistible and non-irresistible infections. Over late years, certain zoonoses, for example, bird influenza or the Ebola and Zika viral scourges, have outlined this reality to the entire world showing the association of human wellbeing, creature wellbeing, and environmental wellbeing. Coming from the "One Health" idea18 that advocates a mix of human medication and veterinary medication because of zoonoses19, the "One World - One Health" concept was made in 2004. The oddity was the joining of environment wellbeing, including that of wild fauna. The "One Health" initiative, in this way, establishes a worldwide technique featuring the requirement for a methodology that is comprehensive and transdisciplinary and joins multisector mastery in managing the strength of humankind, creatures, and biological systems20. At the point when one thinks about the multiple variables at play and the intricacy of general medical problems, unmistakably the "One Health" all encompassing methodology21 can't be disassociated from the idea of environmental wellbeing (EcoHealth). The fundamental reason is that the wellbeing and prosperity of the human populace will be increasingly more hard to keep up with on a dirtied planet experiencing social or political flimsiness and steadily reducing assets.
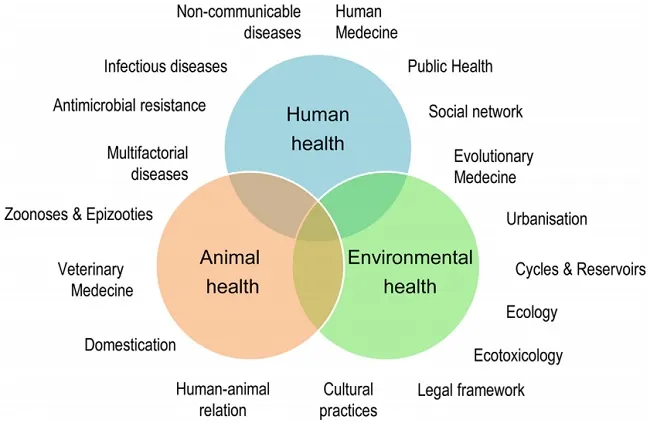
Figure 2: The One Health idea: an all-encompassing, transdisciplinary, and multisectoral approach of Health31.
4.1 Antimicrobial Resistance as a One Health issue:
It is hard to envision an issue that embodies the standards of One Health more than AMR does. The One Health approach, characterized as the communitarian exertion of different disciplines – working locally, broadly, and all around the world – to accomplish ideal wellbeing for individuals, creatures, and our current circumstance27. Perceives that the soundness of individuals is associated with the strength of creatures and the climate. AMR has clear connects to every one of these three spaces. The commitment of creature creation, both earthbound domesticated animals and hydroponics, to the worldwide AMR emergencies is addressed by some in light of the fact that we don't see so many creature related contaminations in humans28. While this might be valid, due to the way that numerous antibiotics are utilized in creature creation, in sub-helpful dosages, and with long openness periods, these creation frameworks make ideal conditions for microscopic organisms to fix qualities that present resistance. These qualities can therefore be sent to human-adjusted microorganisms or to human gut microbiota through individuals, debased food, or the climate. They additionally give ideal conditions to the enhancement of qualities that might have emerged in individuals or the climate. The way that the antibiotics utilized in human and creature wellbeing generally involve something similar or very much like particles would be relied upon to drive the transmission of resistance between creatures and individuals, either straightforwardly or by means of the climate.
Of the three areas, human wellbeing takes the spotlight, with multidrug-resistance qualities now profoundly predominant in numerous significant and normal microbes like Escherichia coli, Klebsiella pneumonia, and Staphylococcus aureus. Very separated from the numerous irresistible infections for which we depend on antibiotics to battle, these medications support present day medication by permitting us to do normal surgeries and therapies that push down the insusceptible framework, like chemotherapy to treat malignant growth. The worldwide weight of AMR on the populace has been estimated29, 30, and more explicit examinations have likewise been done.
5. Discussion
Whatever move we make might be economical in case it depends on a sound understanding of the general jobs of individuals, creatures, and the climate in the rise, spread, and constancy of AMR qualities. This isn't an issue that high-income countries will actually want to tackle alone. The significant job that creature farming plays in vocations in low-and middle-income countries is obscure or disparaged by numerous individuals in high-income countries and should be an indispensable piece of the reasoning and exchange assuming we need to keep away from the entanglements found in environment arrangements, with low-and middle-income countries frequently hesitant to make estimates that might think twice about the transient financial turn of events. The One Health science appears to miss the mark with regards to understanding the monetary powers behind many arising irresistible illnesses; there is a further hole to be spanned between the biomedical, natural, and animal sciences, and the sociologies. Incorporated ways to deal with diminished choice pressing factor and upset AMR transmission cycles on a worldwide scale should be looked for that is established on strong One Health standards as well as in view of financial proof and on standards of social value and worldwide admittance to viable medical care for individuals and their animals.
Acknowledgements
This work was supported through both the industry-academia collaboration project, VtR Inc-CGU, R.O.C., project grant (SCRPD1L0221); and also the CGU project grant (UZRPD1L0011).
6. Reference
1. Bax R, Griffin D. Introduction to antibiotic resistance. InAntibiotic Resistance 2012 (pp. 1-12). Springer, Berlin, Heidelberg.
2. King LJ, Anderson LR, Blackmore CG, Blackwell MJ, Lautner EA, Marcus LC, Meyer TE, Monath TP, Nave JE, Ohle J, Pappaioanou M. Executive summary of the AVMA one health initiative task force report. Journal of the American Veterinary Medical Association. 2008 Jul 15;233(2):259-61.
3. Robinson TP, Bu DP, Carrique-Mas J, Fèvre EM, Gilbert M, Grace D, Hay SI, Jiwakanon J, Kakkar M, Kariuki S, Laxminarayan R. Antibiotic resistance is the quintessential One Health issue. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2016 Jul 1;110(7):377-80.
4. Rousham EK, Unicomb L, Islam MA. Human, animal and environmental contributors to antibiotic resistance in low-resource settings: integrating behavioural, epidemiological and One Health approaches. Proceedings of the Royal Society B: Biological Sciences. 2018 Apr 11;285(1876):20180332.
5. Bryskier, A. (ed.). 2005. Antimicrobial agents: antibacterials and antifungals. ASM Press, Washington, DC.
6. Bro¨tze-Oesterhelt, H., and N. A. Brunner. 2008. How many modes of action should an antibiotic have? Curr. Opin. Pharmacol. 8:564–573.
7. Strohl, W. R. 1997. Biotechnology of antibiotics, 2nd ed. Marcel Dekker, Inc., New York, NY.
8. Nicolaou KC, Montagnon T. Molecules that changed the world. Clinical Pharmacology and Pharmacokinetics. 2008:14.
9. Liu, B., and M. Pop. 2009. ARDB—Antibiotic Resistance Genes Database. Nucleic Acids Res. 37:D443–D447.
10. Morar M, Wright GD. The genomic enzymology of antibiotic resistance. Annual review of genetics. 2010 Dec 1;44:25-51.
11. Randall, C. P., Mariner, K. R., Chopra, I. & O’Neill, A. J. The target of daptomycin is absent from Escherichia coli and other Gram-negative pathogens. Antimicrob. Agents Chemother. 57, 637–639 (2013)
12. Tsuchido, T. & Takano, M. Sensitization by heat treatment of Escherichia coli K-12 cells to hydrophobic antibacterial compounds. Antimicrob. Agents Chemother. 32, 1680–1683 (1988)
13. Kojima, S. & Nikaido, H. Permeation rates of penicillins indicate that Escherichia coli porins function principally as nonspecific channels. Proc. Natl Acad. Sci. USA 110, E2629–E2634 (2013).
14. Vargiu, A. V. & Nikaido, H. Multidrug binding properties of the AcrB efflux pump characterized by molecular dynamics simulations. Proc. Natl Acad. Sci. USA 109, 20637–20642 (2012).
15. Blair JM, Webber MA, Baylay AJ, Ogbolu DO, Piddock LJ. Molecular mechanisms of antibiotic resistance. Nature reviews microbiology. 2015 Jan;13(1):42-51.
16. Gao, W. et al. Two novel point mutations in clinical Staphylococcus aureus reduce linezolid susceptibility and switch on the stringent response to promote persistent infection. PLoS Pathog. 6, e1000944 (2010).
17. Abraham, E. P. & Chain, E. An enzyme from bacteria able to destroy penicillin. 1940. Rev. Infect. Dis. 10, 677–678 (1988).
18. Schwabe C. Veterinary Medicine and Human Health. Baltimore: Williams & Wilkins (1984). 713 p.
19. Zinsstag J, Schelling E, Waltner-Toews D, Tanner M. From “one medicine” to “one health” and systemic approaches to health and well-being. Prev Vet Med (2011) 101:148–56. doi:10.1016/j.prevetmed.2010.07.003
20. One Health Initiative Task Force. One Health: A New Professional Imperative. Schaumburg, IL: American Veterinary Medical Association (2008).
21. Zinsstag J, Schelling E, Waltner-Toews D, Tanner M, editors. One Health, the Theory and Practice of Integrated Health Approaches. CABI (2015). doi:10.1079/ 9781780643410.0000
22. Prescott JF: Antimicrobial drug resistance and its epidemiology. • In Antimicrobial Therapy in Veterinary Medicine, edn 3. Edited by Prescott JF, Baggot JD, Walker RD. Ames: Iowa State University Press; 2000:27-49.
23. Mazel D, Davies J: Antibiotic resistance in microbes. Cell Mol Life • Sci 1999, 56:742-754.
24. Levy SB: The Antibiotic Paradox: How Miracle Drugs are Destroying the Miracle. New York: Plenum Press; 1992
25. Committee on Drug Use in Food Animals, Panel on Animal Health, • Food Safety, and Public Health, Board on Agriculture, National Research Council, Food and Nutrition Board, Institute of Medicine: The Use of Drugs in Food Animals: Benefits and Risks. Washington, DC: National Academy Press; 1999
26. Teuber M. Veterinary use and antibiotic resistance. Current opinion in microbiology. 2001 Oct 1;4(5):493-9.
27. AVMA. One health: A new professional imperative. One Health Initiative Task Force Final Report. Schaumburg, IL: American Veterinary Medical Association; 2008.
28. Chang Q, Wang W, Regev-Yochay G. Antibiotics in agriculture and the risk to human health: how worried should we be? Evol Appl 2015;8:240–5.
29. O’Neill J. Tackling drug-resistant infections globally: Final report and recommendations. The review on antimicrobial resistance; London: HM Government and the Wellcome Trust; 2016.
30. WHO. Antimicrobial resistance: Global report on surveillance. Geneva: World Health Organization; 2014.
31. Destoumieux-Garzón D, Mavingui P, Boetsch G, Boissier J, Darriet F, Duboz P, Fritsch C, Giraudoux P, Le Roux F, Morand S, Paillard C. The one health concept: 10 years old and a long road ahead. Frontiers in veterinary science. 2018 Feb 12;5:14.















