Patient-Centric Approaches in Clinical Trials
Vasudev Bhupathi, Director, Pharmacovigilance, Moderna
Patient-centric clinical trials embed participants’ needs at every stage, co-designed protocols, decentralized visits, digital health tools, diversity metrics, and transparent results. This paradigm accelerates recruitment, boosts retention, enriches real-world data, and builds trust while satisfying evolving regulatory guidance, ultimately delivering safer, more relevant therapies through meaningful patient partnership and measurable value.
Introduction
The traditional clinical trial model has historically prioritized operational efficiency and regulatory compliance, often at the expense of patient convenience and engagement. Traditional site-centric trials typically rely on frequent in-person visits, narrowly defined eligibility criteria, and endpoints focused on clinical markers rather than patient experiences. In contrast, patient-centric trials emphasize convenience, inclusivity, and the measurement of outcomes that matter most to patients.
Recent regulatory updates reinforce this paradigm. The U.S. Food and Drug Administration’s (FDA) Patient-Focused Drug Development (PFDD) guidance series emphasizes systematic collection of patients experience data for trial design and labeling (FDA, 2021). Similarly, the International Council for Harmonisation (ICH) finalized E6 (R3) Good Clinical Practice guidelines in 2025, which call for inclusive design, proportionate oversight, and patient engagement throughout the trial lifecycle (ICH, 2025). Together, these frameworks emphasize that patient partnership is central to modern clinical development.
Patient-Centricity in the Context of Scientific Integrity
The U.S. White House Executive Order on Restoring Gold Standard Science (2025) highlights the need for transparency, reproducibility, and unbiased communication in federally funded science. These principles align directly with patient-centric trials, where trust is established through clear communication of uncertainties, lay-accessible results, and adherence to rigorous safety and ethical oversight (White House, 2025).
By applying these principles, patient-centric trials ensure that participants are not treated merely as data sources but as informed partners, whose involvement strengthens both the scientific and ethical foundations of research.
Key Components of Patient-Centric Clinical Trials'
1. Co-Designed Protocols and Burden Reduction
Patient involvement in trial design ensures that endpoints, visit schedules, and assessments reflect real-world needs and tolerability. Simulation of patient burden, such as travel time, frequency of visits, or procedural invasiveness, allows protocols to be adjusted before trial launch (Dockendorf et al., 2020). For example, the inclusion of patient-reported outcomes (PROs) ensures that quality-of-life dimensions like fatigue or mobility are evaluated alongside clinical efficacy.
Table 1. Comparison of Traditional vs. Patient-Centric Protocol Design

2. Decentralized and Digitally Enabled Models
Decentralized clinical trials (DCTs) leverage telehealth, wearable devices, and home-based sampling to reduce participation barriers. Evidence shows that virtual recruitment can reduce enrollment timelines by 30–40% and improve retention by up to 20% compared with traditional site-based models (Koller et al., 2025). However, digital divides, stemming from inequities in internet access and technology literacy, must be addressed through loaner devices, training, and multilingual support (Aiyegbusi et al., 2024).
Table 2. Tools and Technologies in Decentralized Trials

3. Diversity, Equity, and Inclusion (DEI)
Ensuring diverse representation in clinical trials remains a critical challenge. Patient-centric strategies include prespecifying diversity goals, partnering with community clinics, and tailoring recruitment materials culturally and linguistically (Aiyegbusi et al., 2024). For example, the PREVENTABLE trial successfully increased older adult participation by integrating telehealth visits and home drug delivery.
Table 3. Strategies to Enhance Diversity and Inclusion

4. Clear Communication Pathways
Effective two-way communication is a cornerstone of patient-centric clinical trials, ensuring that participants remain engaged, informed, and motivated throughout the study. Rather than viewing communication as a one-time consent process, patient-centric models emphasize ongoing dialogue between patients and trial teams. This includes the use of plain-language trial materials, accessible support systems, and transparent reporting of study progress.
Effective strategies include:
- Lay-friendly consent documents that make study goals, risks, and procedures understandable.
- Round-the-clock support services such as digital platforms, helplines, or chat-based tools that allow participants to easily ask questions or report issues.
- Regular updates and plain-language summaries of progress and results, reinforcing those participants are true partners in the research process.
Importantly, regulatory frameworks have begun to embed these principles. For example, the European Clinical Trials Regulation (536/2014) requires sponsors to provide lay summaries of clinical trial results, reinforcing the global expectation that transparency and communication are integral to both ethical standards and patient trust.
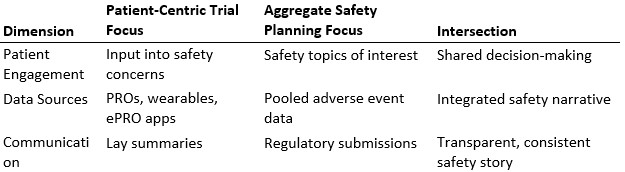
5. Aggregate Safety Planning as a Patient-Centric Safeguard
Aggregate Safety Assessment Planning (ASAP) frameworks, developed through DIA–ASA collaborations, emphasize proactive, interdisciplinary, and continuous safety monitoring across the drug development lifecycle (Hendrickson et al., 2021). Importantly, ASAP integrates patient perspectives when identifying “safety topics of interest,” ensuring that adverse events with quality-of-life impact (e.g., fatigue or mild but persistent side effects) are systematically evaluated (Ball et al., 2019).
For example, patient-reported outcomes (PROs) collected via ePRO apps can be pooled with adverse event reports and laboratory values to provide a more holistic view of safety signals. This integration strengthens benefit–risk assessments and demonstrates to participants that their feedback directly influences trial safety oversight.
Table 4. Intersection of Patient-Centric Trials and Aggregate Safety Planning
6. Regulatory Alignment and Trained Workforce
Patient-centricity cannot succeed without strong adherence to regulatory and ethical frameworks. ICH GCP (E6 R3), FDA’s decentralized trial guidance (2024), and European Medicines Agency’s (EMA) transparency mandates provide the structural backbone for innovation (FDA, 2024; ICH, 2025). Trained staff and active Institutional Review Board (IRB) oversight ensure compliance, data integrity, and participant protection.
Benefits of Patient-Centric Approaches
The transition from traditional, site-centric trials to patient-centric models brings measurable improvements across the clinical research lifecycle. By embedding patient input, leveraging digital health tools, and prioritizing inclusivity, these approaches generate evidence that is both scientifically rigorous and meaningful to participants. Patient-centric designs have demonstrated practical advantages such as faster recruitment, higher retention, improved representativeness of study populations, and greater data relevance. Importantly, they also foster trust through transparent communication and return of trial results, helping to build long-term relationships between patients, sponsors, and regulators.
The following table highlights key benefits identified in recent literature and regulatory guidance, supported by quantitative findings from decentralized and hybrid clinical trial models.
Table 5. Benefits of Patient-Centric Clinical Trials

Challenges and Mitigation Strategies
Despite clear advantages, implementing patient-centric clinical trials is not without challenges. Barriers such as the digital divide, regulatory variability, site workflow disruption, data privacy concerns, and patient adherence fatigue can undermine the success of even the best-designed studies. Addressing these issues requires thoughtful planning, stakeholder engagement, and adoption of enabling technologies that support both patients and trial teams.
Mitigation strategies often include providing technological support to participants, harmonizing processes across jurisdictions, and investing in staff training. Additionally, sponsors are increasingly using gamification and flexible scheduling to improve adherence, while ensuring strict compliance with data privacy standards such as GDPR and HIPAA. The table below summarizes common challenges alongside practical strategies that have been successfully employed to overcome them.
Table 6. Key Challenges and Mitigation Strategies in Patient-Centric Trials

Future Directions
Patient-centric clinical trials are evolving rapidly, driven by advances in digital health, data science, and regulatory support for innovative methodologies. Several emerging directions hold promise for further strengthening patient engagement, data quality, and the overall efficiency of drug development.
Integration of artificial intelligence (AI) and digital twins: AI-enabled tools are increasingly being explored for participant recruitment, predictive modeling, and adaptive trial design. By analyzing large datasets from electronic health records (EHRs), registries, and real-world data sources, AI algorithms can identify eligible patients more efficiently and predict dropout risks in real time. In parallel, the concept of “digital twins”, virtual replicas of patients built from multimodal health data, offers a means to simulate trial outcomes, optimize protocol design, and reduce unnecessary participant burden. Recent FDA workshops highlight AI’s role in ensuring trial representativeness and supporting personalized study pathways.
Tokenized Patient Data Platforms: The shift toward decentralized trials has heightened the importance of secure, patient-controlled data sharing. Blockchain-based tokenization frameworks are being tested to give patients ownership of their health data while enabling sponsors to access high-quality, verifiable datasets. Such platforms can facilitate cross-study data reuse, improve transparency in informed consent, and foster trust by ensuring participants control how and where their data is used. Early pilot projects in Europe demonstrate feasibility for integrating tokenization with remote monitoring and consent workflows.
Real-world Data (RWD) Integration: The incorporation of real-world data into clinical trial design and post-marketing surveillance is gaining traction. Pragmatic and siteless trial designs leverage EHRs, claims databases, and digital health applications to assess treatment effectiveness in naturalistic settings. This approach can reduce costs, shorten development timelines, and enhance the generalizability of findings to diverse populations. Regulatory frameworks such as the FDA’s Real-World Evidence (RWE) program provide guidance on using RWD to complement randomized controlled trials, particularly in rare diseases and post-approval safety monitoring.
Augmented and Virtual Reality (AR/VR) Technologies: Immersive digital technologies such as AR and VR are emerging as tools to improve participant education, adherence, and retention. VR-based modules can enhance patient understanding during the informed consent process, while AR-based reminders and coaching tools support treatment compliance during the study. Additionally, these technologies are being piloted for remote site monitoring and virtual investigator meetings, which could reduce logistical burdens and improve trial accessibility for geographically dispersed populations.
Together, these innovations illustrate a future in which clinical trials are not only more efficient and scientifically rigorous but also deeply aligned with patient needs and experiences. By combining advanced technologies with patient-centric design principles, the industry has an opportunity to fundamentally transform the evidence-generation ecosystem.
Conclusion
Patient-centric approaches are reshaping clinical development by embedding participants’ perspectives throughout the trial lifecycle. Grounded in regulatory expectations, safety frameworks, and principles of scientific integrity, these models accelerate recruitment, enhance retention, improve equity, and build trust. By integrating patient input with robust safety and ethical oversight, clinical research can deliver therapies that are safer, more relevant, and more meaningful to the people they are designed to serve.







