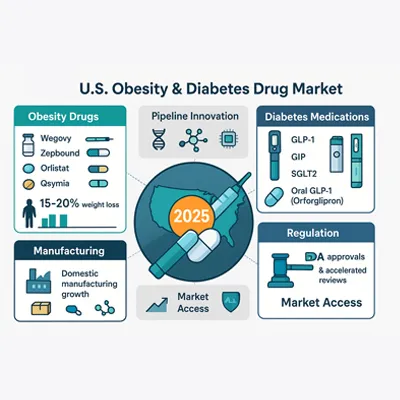
Top Obesity and Diabetes Medications Transforming U.S. Pharma Market
Introduction: U.S. Obesity and Diabetes Drug Market Trends
The U.S. healthcare system is at a turning point as the dual epidemics of obesity and type 2 diabetes drive demand for advanced treatments. More than 42% of American adults are obese, while over 37 million people live with diabetes. These conditions are not only straining public health but also reshaping the pharmaceutical industry, creating a booming market for anti-obesity drugs and diabetes medications.
In 2025, pharma companies are witnessing unprecedented growth in the obesity drug market, with GLP-1 receptor agonists leading the way. The demand for FDA-approved weight loss drugs and next-generation diabetes treatments is fueling innovation, manufacturing expansion, and regulatory scrutiny across the U.S.
Anti-Obesity Medication and Diabetes Medication Market Growth in the U.S.
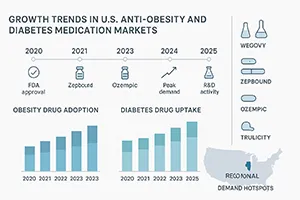
The increasing rates of obesity and diabetes in the U.S. have created an urgent need for advanced medical treatments, fueling rapid growth in both the anti-obesity medication and diabetes medication sectors. According to recent market analyses, the obesity drug market growth is expected to accelerate through 2030, reflecting expanded awareness and improved drug efficacy.
Pharmaceutical companies are aggressively investing in research and development, expanding the diabetes drug pipeline with novel therapies designed to manage blood sugar levels and promote weight loss. This robust pipeline includes drugs that not only target diabetes but also address obesity, recognizing the close link between the two conditions.
Novo Nordisk Obesity Drugs and Eli Lilly Diabetes Medications Leading the Market
Two pharmaceutical giants, Novo Nordisk and Eli Lilly, dominate the U.S. market with their innovative portfolios of obesity drugs and diabetes medications. Novo Nordisk’s blockbuster product, Ozempic, a GLP-1 agonist, has revolutionized both diabetes and weight management treatments. Meanwhile, Eli Lilly’s Trulicity and newer diabetes drugs continue to gain traction for their effectiveness and patient adherence.
These companies have extensive networks of pharmaceutical distributors for diabetes medications, ensuring that their products reach healthcare providers and patients nationwide. Their leadership in this space not only highlights the commercial potential but also underscores the critical role these drugs play in combating the obesity epidemic.
Role of GLP-1 Agonists in Diabetes Treatment and Anti-Obesity Medication
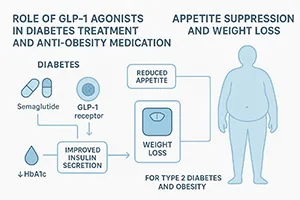
One of the most groundbreaking developments in recent years is the rise of GLP-1 agonists. These drugs mimic the glucagon-like peptide-1 hormone, which regulates appetite and insulin secretion. The dual action of these medications makes them highly effective in controlling blood glucose levels while simultaneously promoting weight loss.
Understanding how GLP-1 agonists are transforming diabetes treatment and pharmacotherapy for obesity is essential for industry stakeholders. Beyond their clinical benefits, these drugs represent a strategic advantage for pharmaceutical companies aiming to address two major health crises with a single therapeutic class.
Emerging Obesity Drugs and Diabetes Drug Pipeline 2025 in the U.S. Pharma Market
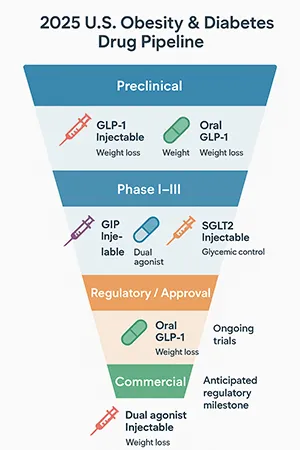
Looking ahead, the emerging obesity drugs in pharmaceutical pipeline 2025 are expected to further disrupt the market. Several novel compounds and combination therapies are undergoing clinical trials, targeting different metabolic pathways to enhance efficacy and safety.
The evolving diabetes drug pipeline also includes innovative agents designed to reduce cardiovascular risks and improve quality of life for patients. Ongoing clinical trials for new diabetes drugs are critical to expanding treatment options and maintaining momentum in the fight against diabetes and obesity.
Top FDA-Approved Obesity Medications Reshaping Pharma
The anti-obesity drug market is one of the fastest-growing segments in U.S. pharma. Key FDA-approved weight loss drugs in 2025 include:
- Wegovy (semaglutide): A GLP-1 analog offering significant weight reduction.
- Zepbound (tirzepatide): A dual GIP/GLP-1 agonist with superior efficacy in trials.
- Orlistat (Xenical, Alli): A fat absorption inhibitor still in use but less favored.
- Qsymia (phentermine-topiramate): Effective but facing adoption challenges.
These drugs are transforming patient care while intensifying competition among pharmaceutical companies.
Latest Advances in Diabetes Treatments and Market Expansion
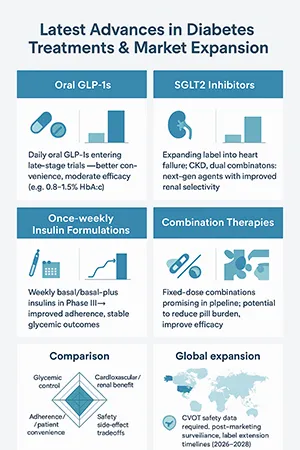
The diabetes drug pipeline is equally dynamic. Type 2 diabetes treatments in 2025 go beyond insulin therapy, with emphasis on oral GLP-1s, SGLT2 inhibitors, and once-weekly insulin formulations.
Notable innovations include:
- SGLT2 inhibitors improving both glycemic control and kidney health.
- Once-weekly insulin analogs simplifying adherence.
- Combination diabetes therapies targeting multiple pathways for better outcomes.
For pharma companies, these advancements open new opportunities in formulation development, manufacturing partnerships, and global expansion.
Oral GLP-1 Medications: Orforglipron and Beyond
The next frontier is oral GLP-1 medications like Orforglipron, developed by Eli Lilly. Unlike injectable GLP-1s, Orforglipron offers daily oral dosing, improving patient adherence and accessibility.
Clinical trials show up to 12.4% weight loss over 72 weeks in non-diabetic patients, positioning it as a potential blockbuster. As more oral formulations enter the pipeline, the pharma market must prepare for scale-up manufacturing and supply chain demands.
Comparing Semaglutide vs. Tirzepatide: Efficacy and Market Positioning
One of the most debated comparisons in 2025 is Semaglutide (Wegovy) vs. Tirzepatide (Zepbound).
| Feature | Semaglutide (Wegovy) | Tirzepatide (Zepbound) |
| Mechanism | GLP-1 agonist | Dual GLP-1/GIP agonist |
| Avg. Weight Loss | ~15% body weight | ~20% body weight |
| Administration | Weekly injection | Weekly injection |
| Market Share | High, but challenged | Rapidly expanding |
For pharma stakeholders, this head-to-head competition highlights opportunities in market differentiation, patient segmentation, and clinical positioning.
Pharmaceutical Manufacturing and Supply Chain Challenges
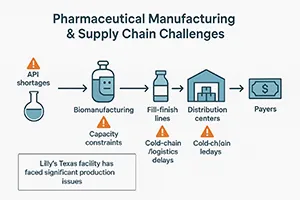
With rising demand for obesity and diabetes drugs, manufacturers face significant challenges:
- API shortages due to limited global supply.
- Production capacity constraints with injectable biologics.
- Distribution bottlenecks in meeting U.S. demand.
Eli Lilly’s $6.5B Texas manufacturing facility demonstrates the industry’s urgent need for domestic ingredient production. For suppliers, contract manufacturers, and technology providers, this presents a strong B2B growth opportunity.
Regulatory Landscape for Obesity and Diabetes Medications
The FDA approval process is under pressure as obesity and diabetes drugs dominate clinical trials. Key regulatory themes include:
- Accelerated approvals for high-demand therapies.
- Safety and cardiovascular monitoring requirements.
- Pricing and reimbursement debates with payers.
Pharma companies must carefully navigate this landscape to ensure timely launches while meeting compliance standards.
Market Access Strategies and Pharmaceutical Innovations in Diabetes Care and Obesity Drug Market
Securing market access remains a complex challenge for pharmaceutical companies. Effective obesity medication market access strategies involve navigating regulatory approvals, reimbursement policies, and payer negotiations. Companies that successfully implement these strategies can rapidly scale their presence in the competitive U.S. market.
Moreover, continuous pharmaceutical innovations in diabetes care—including digital health integrations and personalized medicine—are revolutionizing patient management. Collaborations and pharma partnerships for obesity drug development are accelerating innovation, combining expertise to bring next-generation therapies to market faster.
Future Outlook: Innovations in Obesity and Diabetes Treatments
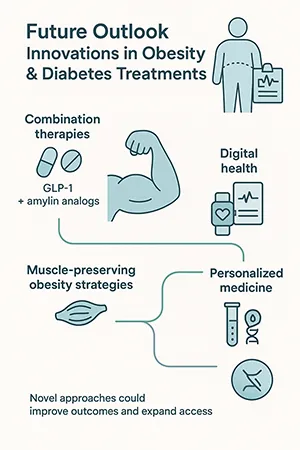
The future of obesity and diabetes care will likely include:
- Combination therapies (GLP-1 + amylin analogs).
- Muscle-preserving obesity treatments to counter lean mass loss.
- Digital therapeutics integration with drug regimens.
- Personalized medicine approaches driven by genetics and biomarkers.
Pharma companies that embrace innovation, partnerships, and patient-centric strategies will lead the next era of healthcare transformation.
Conclusion: How Obesity and Diabetes Medications Are Transforming U.S. Pharma
The rise of GLP-1 receptor agonists, oral obesity drugs, and advanced diabetes treatments is redefining the U.S. pharma market. Beyond patient outcomes, these therapies are driving investment, regulatory shifts, and manufacturing expansion.
For pharma stakeholders, the message is clear: the obesity and diabetes drug market is not just a therapeutic opportunity, but a strategic growth engine for the next decade.