Twin-Screw Melt Granulation: A Solvent-Free, Single-Step Solution for Enhancing Drug Solubility
Dinesh Nyavanandi, Research Scientist, Drug Product Design and Development, Cerevel Therapeutics
Nithin Vidiyala, Principal Scientist, Drug Product Design and Development, Cerevel Therapeuties
Twin-screw melt granulation (TSMG) is a solvent-free, single-step, and scalable technology enhancing solubility and bioavailability of poorly water-soluble APIs. By improving dissolution, process efficiency, and patient-centric product design, TSMG supports sustainable and continuous manufacturing. This article highlights its fundamentals, advantages, applications, and future role in pharmaceutical innovation.
Over the last few decades, the pharmaceutical industry has been facing persistent challenges in delivering poorly soluble drug candidates. Every pipeline has promising drug candidates that are stalled by the solubility limitations. In recent years, the twin-screw melt granulation (TSMG) approach has been most widely studied and evolving as a promising solution for solubility enhancement.
The solubility challenge
Oral dosage forms are widely preferred by the patient population, attributing to self-administration and low cost. Drug candidates being administered orally should possess good aqueous solubility to be dissolved at the physiological pH and absorbed into the systemic circulation. Around 60-80 % of the pipeline candidates are claimed to be poorly soluble, belonging to the biopharmaceutical classification system (BCS) class II (Poor Solubility and High Permeability) and IV (Poor Solubility and Poor Permeability). Failure to enhance solubility and oral bioavailability results in the selection of an alternate dosage form, such as injectables. Even a few drug candidates are discontinued from the clinical evaluation due to low efficacy.
Traditional approaches such as micronisation, co-crystal, salt, and complexation have resulted in improved solubility but involve the use of solvents and multiple unit operations. In the current landscape where cost-effectiveness and efficiency are paramount, there is a need for the development of robust and efficient manufacturing processes.
Twin screw melt granulation (TSMG)
The TSMG process is carried out using a traditional hot melt extruder (HME) with slight modifications. The TSMG process involves feeding material consisting of drug and excipients with a low-melting-point binder into the extruder barrel. The barrel consists of a pair of co-rotating screws with different zones, namely “conveying” and “mixing” zones. The screw configuration can be adjusted using conveying, kneading, and comb mixing elements. The conveying elements mainly transfer the material between the zones but do not possess a mixing property. The kneading elements can be adjusted to a different offset angle, where, with the increasing angle, the amount of shear being imposed on the processing material increases. The kneading elements will impose dispersive and distributive mixing on the processing material. The material fed into the barrel gets exposed to thermal and mechanical shear. The conveying elements transfer the material from one zone to another. In the TSMG process, at the processing temperature, a low melting point solubilising excipient gets melted and dissolves the drug substance and adsorbs onto the solid carrier materials such as microcrystalline cellulose, lactose, and mannitol, etc. The comb mixing elements will aid the size reduction of large-sized granules, eliminating the need for a downstream milling operation. Following the comb mixing elements, the granules are conveyed and collected at the end of the barrel. The critical process parameters that are involved within the TSMG process include feed rate, barrel temperature, screw speed, and screw configuration. A detailed instrumentation of the TSMG is shown in Figure 1.
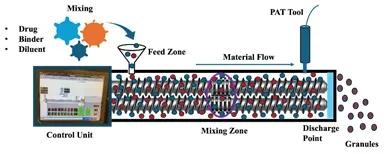
Figure 1: Detailed instrumentation of TSMG equipment
Advantages of TSMG
Implementation of the TSMG process provides a unique combination of benefits.
● Solvent-free: Compared with traditional manufacturing, the TSMG can be considered as “Green Technology” with no need for solvents. This also eliminates the downstream drying step, making the process more efficient and cost-effective.
● Single-step process: The traditional manufacturing approaches involve multiple unit operation steps, which are time-consuming. The TSMG is a single process that consists of melting, mixing, and granulation, reducing the number of processing steps with small-footprint equipment.
● Solubility and permeability enhancement: Within the TSMG process, the solubilising carrier can be melted to dissolve the drug and can transform it into an amorphous form. Along with other carrier materials that exist in dry form, a permeability enhancer can also be added, which plays an important role in enhancing the bioavailability of BCS class IV drug candidates, along with class II molecules.
● Scale-up: Within small molecule drug development, the scale-up of the manufacturing process is more critical. The traditional manufacturing involves optimisation of multiple unit operations, each with various critical process parameters. The scale-up of the TSMG process is straightforward. In fact, the r&d scale equipment used for the developmental work can also be used for manufacturing with the same process conditions, extending the run time.
● Continuous manufacturing: In recent years, the industries and regulatory agencies have become interested in the continuous manufacturing process. The TSMG requires a single piece of equipment for the granulation process, which can be integrated with downstream instruments such as encapsulation and tablet compression to establish a continuous manufacturing line. In fact, suitable process analytical technology tools (PAT) can be implemented for continuous monitoring of the manufacturing process and quality of the product. In case of any discrepancy, the process can be stopped, saving unprocessed material and reducing the wastage.
For the pharmaceutical industries, all the advantages being offered by TSMG translate into shorter processing time, lower cost, and a high success rate for advancing the challenging drug candidates within the pipeline.
Industry and regulatory perspective
The implementation of the emerging technology program by the Food and Drug Administration (FDA) and the ICH Q13 guideline for continuous manufacturing shows the interest and encouragement of the regulatory bodies to shift from batch to continuous manufacturing. The TSMG process, being a single-step, solvent-free process, matches the regulatory expectations. By eliminating the need for solvent and intensive drying steps, the TSMG process provides the opportunity for the pharmaceutical industries to push towards “green manufacturing”. As more companies adopt the TSMG approach, the regulatory bodies will view it as a feasible technology rather than limiting it to an experimental process.
Challenges and future outlook
Similar to other traditional manufacturing techniques, the TSMG also has limitations. The application of thermal energy is the primary concern limiting the feasibility of this process for heat-sensitive drug candidates. There is a need for developing novel excipients that can be processed at low temperatures, making the platform suitable for thermal sensitive drug molecules. Another limitation is the processing of drug candidates that are sensitive to mechanical shear. For the processing of materials sensitive to mechanical pressure, the screw configuration of the mixing zones needs to be optimised to ensure application of low shear on the processing material. Implementation of advanced PAT tools, such as near infrared (NIR) and Raman, enables real-time monitoring of the product quality. In the future, the TSMG process will play an essential role in the implementation of a continuous manufacturing platform.
Conclusion
Overall, the TSMG process has resulted in a powerful evolution within the pharmaceutical sector by providing the flexibility of a single-step, solvent-free, and easily scalable process. The TSMG approach can be successfully integrated into the continuous manufacturing platform, providing the pharmaceutical industries with an opportunity to switch from the traditional batch manufacturing process.











