Data Exchange in Pharmaceutical Manufacturing
Balancing Collaboration and Competition
John Ward, Founder and CEO of ServBlock
Mick Cummins, Managing Director of Ingeniero Solutions
The Challenge of Managing Data, in Pharmaceuticals; Balancing Collaboration and Competition" delves into the equilibrium between sharing data and maintaining an edge within the pharmaceutical industry. This article explores the obstacles and advantages associated with manufacturing examining how the exchange of proprietary information impacts innovation, efficiency and adherence to regulations. It underscores the importance of data security measures and ethical sharing practices showcasing real life examples where successful collaboration resulted in expedited drug development and production. Ultimately this piece serves as a resource, for stakeholders navigating the realm of pharmaceutical manufacturing partnerships.

In the pharmaceutical industry, the importance of sharing proprietary information plays a crucial role in driving innovation. Developing new drugs and therapies is a complex and time consuming process that can be significantly expedited through data sharing. Collaborative efforts, such as joint research initiatives or platforms for sharing data, have the potential to accelerate the discovery of new drug candidates and treatment methods. Similarly, when sponsor companies outsource manufacturing to Contract Manufacturing Organizations (CMOs), they need to be willing to share trade secrets in order to maximize productivity, ensure product quality and minimize time before patients can benefit from these medications. However, concerns about intellectual property theft or loss of exclusivity may discourage companies from engaging in open collaborations, thereby potentially slowing down the pace of innovation and manufacturing output.
Sponsor companies that own drug formulas rely on CMOs to manufacture their pharmaceutical products while maintaining the same level of quality assurance as if they were producing them in house. This collaboration requires sharing essential information for the manufacturing process. CMOs play a vital role by utilizing their production capabilities and expertise to produce drugs at scale while adhering to quality standards and compliance regulations.
However, the mutually beneficial relationship between sponsor companies and CMOs becomes complex when it comes to managing sensitive proprietary data. If this data is compromised, it could have serious financial and reputational consequences for both parties involved.
In this article, we will delve into the intricacies of data management within the context of sharing information between sponsor companies and CMOs. We will explore the challenges and opportunities that arise when sponsor companies outsource their operations to CMOs. Our focus will be on understanding the extent of data exchanged between these entities, addressing the associated risks related to data security and exploring available options to mitigate these risks.
The Challenge
Sharing information plays a crucial role in driving innovation in drug manufacturing. When sponsor companies collaborate with Contract Manufacturing Organizations (CMOs), they can leverage the capabilities and technologies offered by these organizations. This collaboration aims to bring about advancements in drug manufacturing processes, enhance cost efficiencies and reduce time to patient.
However, outsourcing Good Manufacturing Practice (GMP) processes like this requires rigorous oversight by the sponsor company throughout the entire lifecycle of the process at the CMO manufacturing facility. This oversight is made possible through data exchange between sponsors and CMOs. It allows sponsors to evaluate various aspects such as batch manufacture performance, batch release documentation, deviations in manufacturing processes, raw materials analysis, supply chain information and more.
Sharing real time production data is highly advantageous in refining manufacturing processes, ensuring the production of drugs that meet efficacy and safety standards. It helps save valuable time when scaling up production, which is crucial in an industry where every day counts. Furthermore, CMOs may possess production techniques that improve drug quality or minimize waste during manufacturing. By accessing such data, sponsor companies can incorporate these advancements into their own products, resulting in better outcomes for end users and potentially accelerating the time to market for new drugs.
Regrettably, the pharmaceutical industry often relies on outdated methods of data exchange such as emails, spreadsheets and physical documents. These manual processes come with inherent challenges and risks. Sharing information through email or spreadsheets can lead to loss, misdirection or interception of confidential data while also potentially corrupting it. Such methods necessitate time consuming manual handling of data that is prone to errors due to the effort required for organization, sending and tracking. As the volume of data increases, these manual processes become increasingly burdensome and inefficient. Additionally, the absence of real time sharing capabilities can cause delays in decision making.

Data security is another critical aspect within the lifecycle of CMOs and sponsor companies that demands proper attention.
The concern over the public disclosure or unauthorized access to data by competitors is a significant issue. Intellectual property plays a vital role in the pharmaceutical industry and any leaks without permission could give competitors an advantage or even lead to losing market exclusivity. Therefore, it is essential to find a balance between sharing data for effective collaboration and protecting valuable intellectual property. To achieve this, it is crucial to use protocols that safeguard shared data through secure electronic transfer systems and controlled access measures. Regular audits should be conducted to ensure both parties adhere to the agreed upon standards of data management.
Fortunately, advancements in data management technology now enable secure sharing of information between organizations. These technologies streamline the transfer of sensitive data from one site to another while maintaining stringent security protocols for its protection.
The Evolution of Data Sharing Technologies
The field of data management is evolving alongside the introduction of innovative technologies like data spaces and unified name spaces. These concepts are more than mere buzzwords; they form part of an emerging framework designed to empower those who generate data by allowing them not only to share it but also control how it is consumed and used by their supply chain partners.
In this article, we will explore the practical applications of these concepts in various scenarios such as deviation management, troubleshooting, real time in process control monitoring, quality control (QC) results and investigations within the relationship between CMOs and sponsor companies.
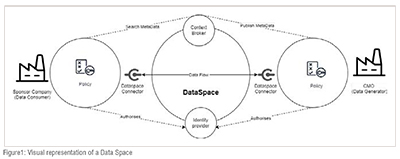
Data Space
In the realm of managing data in today's world, several essential technical components play crucial roles in ensuring the integrity, security and efficient utilization of data. Firstly, we have the concept of a 'Data Space,' which refers to a structured digital environment that stores, processes and manages data from various sources. It's a scalable and flexible framework typically based on cloud technology that allows for the integration and compatibility of diverse datasets. Within this space, 'Identity Management' becomes a vital aspect involving processes and technologies that authenticate and authorize individuals and systems to access specific data. This ensures that each entity interacting with the data space is uniquely identified and granted access rights according to predefined policies.
Data spaces serve as central hubs that enable multiple stakeholders to securely share and manage data within a governed environment. Data spaces act as central hubs where data creators have control over who can access their data, how it is used and the conditions under which it is utilized.
It's worth noting that data spaces can be designed to adhere to important standards like GDPR or HIPAA—which are vital within the pharmaceutical industry. Secure data sharing not only reduces access risks but also ensures compliance with industry regulations. (Figure 1)
Data Sovereignty
When it comes to business, data sovereignty refers to a company's ability to retain control over its data regardless of where it is stored or processed. It plays a crucial role in data management by ensuring that valuable company information remains within the organization's boundaries and is used in alignment with its values, regulatory requirements and business objectives.
One effective approach to strengthen data sovereignty is through the implementation of “Policy Enforcement” within the data space. 'Policy Enforcement' encompasses setting up and managing rules and protocols governing how data is accessed, manipulated and shared within the data space.
Enforcing policies is crucial to ensure adherence to legal regulations, industry standards and internal guidelines. This usually entails the use of automated systems that oversee and regulate data interactions, ensuring that all activities within the data realm align with established policies. These components collectively establish a robust foundation for managing data in todays intricate and data centric environments.
Context Broker
Another key component is the 'Context Broker.' Acting as an intermediary layer within the data space, it collects and analyzes contextual information from different sources. By understanding relationships between various data points, it enables more intelligent processing of information tailored to specific situations. This aspect holds great significance for applications such as smart cities or IoT environments where real time processing and decision making are critical.
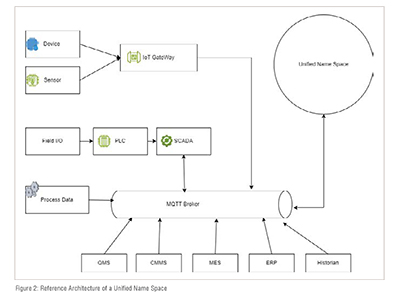
Unified Namespace (UNS)
The concept of a UNS holds immense significance in industrial automation and data management, including within pharmaceutical manufacturing. It is revolutionizing how these industries operate by centralizing and standardizing data. The UNS serves as a centralized hub that collects and stores information from various sources involved in the manufacturing process. This encompasses sensor generated information from the distributed control system (DCS), equipment related details from a computerized maintenance management system (CMMS), as well as system inputs from ERP (Enterprise Resource Planning), MES (Manufacturing Execution System), QMS (Quality Management System) and LIMS (Laboratory Information Management System). By having a single point of access, it simplifies overall data management.
Unified name spaces establish an organized system for naming and retrieving information across different systems, making it easier to locate and access relevant data.
Just imagine navigating through a network of databases where each one has its own unique way of naming files. It can be quite complicated, right? Well, that's where a UNS comes in to simplify things. It's like having a common language specifically designed for retrieving information, making it easier to find what you need. This is particularly useful in troubleshooting scenarios, as engineers and technicians can quickly locate manufacturing information across different companies using this unified referencing system. It saves time and reduces errors.(Figure 2)
The Technological Building Blocks for Balancing Collaboration and Competition
Now, let's talk about the benefits of combining a UNS with a data space in the context of contract pharmaceutical manufacturing and its relationship with the sponsor company. This integration brings clear advantages such as enhanced collaboration, improved transparency and more effective decision making based on data.
In practical terms, the data space allows for controlled sharing of information between the contract manufacturer and the sponsor company. This seamless exchange ensures that both parties are aligned when it comes to production processes, quality control measures, quality assurance practices, supply chain requirements and timelines and technology transfers.
Examples of potential use cases for such data sharing technology in the area of pharmaceutical contract manufacturing are listed below. For these use case examples, data exchange is typically done in fragments through emails, drop boxes and sharepoints, among other methods. However, having a shared dataspace that brings together all the information related to a manufacturing process from various data sources within a manufacturing facility, with appropriate security measures in place, would eliminate the need for manual and inefficient knowledge transfer processes that are commonly observed in the pharmaceutical industry.
Real Time Process Analysis
By utilizing a data space, the sponsor company gains real time access to CMO manufacturing process data. This level of transparency enables monitoring of production activities to ensure compliance with quality standards while ensuring the process is operating within established boundaries and meeting regulatory requirements.
Deviation Management
When there is a deviation from operating procedures or filed/qualified process parameters, it is important to understand why it occurred. Data spaces facilitated by context brokers allow controlled sharing of essential investigative data such as batch records, process control trending, historical deviations within the same system or category, machine logs and environmental monitoring data with external stakeholders. By granting access to this data, teams can effectively collaborate in identifying the root cause behind deviations without compromising data security.
Change Management
Sponsor companies will have on demand access to review change controls that impact regulatory filing of qualified processes at the CMO facility. Ease of access to such information enables and expedites informed decision making at sponsor company leadership level and ultimately will lead to a much speedier progression of the CMO change control process.
Supply Chain
Having a shared data space improves supply chain optimization. Both parties involved can manage raw material and consumable inventory, production schedules and logistics efficiently, resulting in fewer delays and cost savings.
Technology Transfer
Shared data spaces facilitate efficient and robust technology transfers between the sponsor company and the contract manufacturing organization (CMO) or vice versa. When introducing a new product at either facility, it is crucial to have comprehensive knowledge transfer between sites.
Equipment Reliability
When there are issues with the equipment that affects the production process at the CMO, the Sponsor Company may want to conduct a joint investigation to determine the root cause. Engineers can make use of a data space that facilitates seamless data sharing across different companies within the CMO and Sponsor relationship. This allows for easier identification of problems and potential solutions.
Quality Control
QC testing and its results are highly sensitive and prone to human error. In cases where unusual or out of specification results occur, particularly in instances that affect batch release, a sponsor company may wish to have oversight on the testing methods and associated documentation. To ensure authorized personnel can access QC data and any anomalies are investigated, a context broker can be used to facilitate machine to machine interactions within the Dataspace.
Conclusion
To sum up, the concepts of Data Spaces, The Unified Name Space and context brokers all contribute to improving data sharing practices while maintaining data ownership and ensuring ethical handling. These technologies give data creators control over how their data is used while also ensuring integrity. As these technologies become more integrated into data management practices, they have the potential to significantly enhance efficiency and security in operations involving data.
It should be a goal of all pharmaceutical sponsor companies to embrace this new approach to data sharing, particularly inter-organisational data sharing with CMOs. As the industry moves forward with advancements in technology and more stringent regulations, it becomes increasingly important for sponsor companies and CMOs to consistently enhance their practices of sharing data. By doing so, they can foster innovation, maintain operational efficiency, and uphold the standards of data security and ethical responsibility.








