Peptide Therapeutics and Vaccines
New Forms of Delivery and Drug Development
Samatha, Editorial Team, Pharma Focus America
There has been an extremely high revival in the development of the peptide-based drugs because of the natural properties that were available in the peptides, these are the high target specificity, biocompatibility and the flexibility of their structure. The newer trends in delivery platforms (nanocarriers, hydrogels and conjugation strategies) are crucial in boosting the pharmacokinetic performance of peptides that have long been plagued by weak stability and fast degradation. The developments have placed peptides at the epicenter of the therapeutic and vaccine design approaches against a broad range of diseases, including oncology, metabolic, neurological, and infectious. This is an overview of the recent advances in the field of peptide-based drug delivery, therapeutic use of peptide drugs, and even the new implications of peptide-based vaccines, pointing out achievements of the field and open issues.
The importance of peptides, short sequences of amino acids connected with peptide bonds, is also based on their strong biological activity and great affinity to targets and acceptable safety in general. In contrast to mol-based drugs that tend to have off-target and toxic effects, peptides have potential to act on particular signaling pathways and protein-protein interactions in a very specific fashion. They provide an intermediate position between small molecules and biologics and thus fill size, complexity, and manufacturing gaps.
In spite of these strengths, the track record of peptide therapeutics has traditionally been limited by a number of pharmacological obstacles, especially low metabolic stability, high renal clearance, and low Oral bioavailability. Recent renewed interest in the peptide drugs can be credited to great advancements in the field of medicinal chemistry, drug delivery technologies, and peptide engineering. Even more these advances have not only reduced the classic shortcomings, but have allowed the rational design of peptides with tailor-made pharmacokinetics and pharmacodynamics.
This paper outlines the contemporary state of the art of the peptide-based drug development taking into account the state of the edge delivery systems, therapeutic innovations, and vaccine approaches. It is highlighted that multidisciplinary methods of integration are necessary to overcome the current drawbacks and to increase the clinical potential of peptide-based modalities.
The Potential and the Burden of Peptide Therapeutics
The distinct characteristic of designing drugs using peptides is that the latter are of endogenous origin or in other words, they are highly biocompatible and low immunogenic. In fact, they are very useful in targeted treatment because they can bind against specific cellular targets, including receptors, and also against enzymes. The therapeutic peptides may be formulated in such a way that they can be receptor agonists or antagonists, enzymes inhibitors, or even as substrates that influence physiological functions.
Clinical success of glucagon-like peptide-1 (GLP-1) receptor agonist in the 0management of type 2 diabetes/obesity is one example. Bortezomib which is a proteasome inhibitor is an example of peptide used oncology, whereas others focus on cardiovascular and stomach diseases. There are, however, inherent problems. Peptides also have short plasma half-lives that are prone to enzyme degradation by digestive enzymes of the GI tract and blood proteases. They are also hydrophilic and large to passively diffuse through cell membranes.
Among methods to circumvent these obstacles are structural changes (e.g. cyclization, insertion of D-amino acids), protective carrier design, and the combination with enzyme inhibitors. Nevertheless, delivery continues to be one of the significant bottlenecks that require the development of novel platforms that would improve the peptide bioavailability and stability.
Peptide Drug Delivery Platforms
Maximizing therapeutic opportunity of peptides centered on efficient delivery systems. The subsequent passages discuss the recent trends in this area.
Nanocarriers Di lipid-based Systems
The use of lipid-based delivery systems has received a lot of fame because they encapsulate and safeguard peptide drugs. Commonly used mRNA vaccine delivery material: Lipid nanoparticles (LNPs) have been widely used to reorganize transport peptides. They allow a biocompatible support that protects peptides against enzymatic breakdown and gives them the possibility to release controlled delivery.
Bilayered vesicles formed of phospholipids (liposomes) are widely examined as potential vehicles to encapsulate peptides. Advanced features are provided by the solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs), the features of which are enhanced cellular uptake, low toxicity, and physical stability. Such systems may be made target-specific through modification at the surface e.g. with an antibody.
Polymeric Delivery Machinery
The common methods of peptide delivery through biodegradable polymers include: poly (lactic-co-glycolic acid) (PLGA) and many others. Sustained release and site- specific targeting are possible with these polymers. The polymer matrix may be designed to release the payload in a controlled fashion (in response to pH, temperature, or the presence of enzyme activity).
Cell Penetrating Peptides (CPPs)
CPPs are small peptides which can be able to transport molecular cargos, carrying therapeutic peptides, into cells. Such vectors usually are modifications of naturally occurring proteins like HIV-Tat or penetratin. CPPs either operate by momentarily breaking the lipid bilayer or through endocytic routes.
The recent studies aim at making CPPs more efficient and less cytotoxic. CPP-peptide conjugates have held potential to target the previously undruggable intracellular proteins, including transcription factors, kinases.
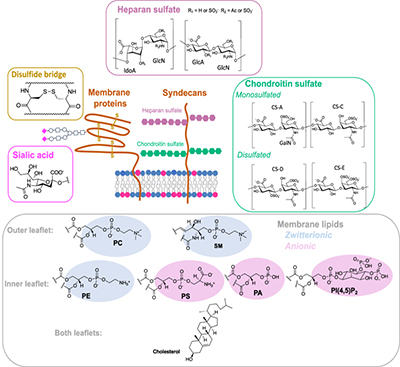
Hydrogels and Microneedles
Hydrogels provide a flexible platform of local and protracted delivery of peptides. These hydrophilic networks of polymers can either be injected or implanted, and develop an in situ depot that releases peptides gradually with time. Hydrogel mechanical quality and deterioration rate can be modified so as to meet certain medical demands.

Microneedle patches form an option that is less invasive to use in transdermal delivery. Microneedles can penetrate through the stratum corneum and offer a pain-free system of effective peptide absorption due to their ability to deliver the peptide. They are especially favorable in vaccine delivery, providing benefits of possible self-administration and increased stability of storage.
Peptide Therapeutics: Applications and Innovations
Oncology
Peptide strategies employed in cancer therapy involve various modalities, namely, targeting receptors, blocking protein-protein interactions, and targeting the delivery of cytotoxic agents. Homing tumor peptides, including integrin targeting RGD integrating motifs, increase the specificity of the treatment delivery, minimizing off-sites.
The use of peptide-drug conjugates (PDCs), similar to antibody-drug conjugates (ADCs), incorporates targeting peptides with chemicals used in chemotherapeutics. These are constructs that take advantage of the overexpression of receptors on tumor cells allowing them to be selectively internalized. Immune check-point modulators and example peptide vaccines that are being studied to promote anti-tumor immunity include the following.
Metabolic Disorders
The example of success of the peptides in the treatment of metabolic diseases is the GLP-1 receptor agonists such as liraglutide and semaglutide. These peptides promote the release of insulin, inhibition of glucagon secretion and curtail the appetite. Their half-life and ease of dosing have been enhanced by modification, e.g. acylation or PEGylation.
New targets are glucose-dependent insulinotropic polypeptide (GIP) and amylin with dual or triple agonists being more efficacious. Delivery systems are under development which would allow oral or one dose per week administration, in order to enhance the adherence and the therapeutic outcome.
Infectious Diseases
Antimicrobial peptides (AMPs) belong to the innate immune response with broad-spectrum specificity among bacteria, viruses and fungi. They induce disruption of the membranes of microbes as well as able to regulate host immunity. AMP mimetics and synthetic AMPs are in evaluation in the clinic based on multidrug-resistant infections.
Viral inhibitors have been created based on the use of peptides to prevent viral infections (HIV, influenza and SARS-CoV-2). Issues are maximizing stability and preventing resistance. The use of AMPs in combination with conventional antibiotics could provide synergistic benefits and slow development of resistance.
Neurological Disorders
Peptidic neurotherapeutics inhibit various mechanisms, such as, inhibition of amyloid-beta aggregation in Alzheimer regional medicine hospital disease, regulating neurotrophic factors, disrupting pathogenic protein interactions. Transport barriers over blood-brain barrier (BBB) exist as a major challenge.
Ways to overcome this are conjugation with CPPs or receptor mediated transcytosis ligands. Another approach to administration that is actively pursued is intranasal routes of administration that do not cross through the BBB. In neurodegenerative and psychiatric disorders, there is asset in a study ongoing in clinical and unlicensed translation.
Mechanisms and Usage of Peptide Vaccines
Invention and Conveniences
Peptides vaccines utilize small size (synthetically made), antigen-specific epitopes in order to create immune responses. They present some benefits: they are defined, safe, stable, and easy to generate. The peptide vaccines help to reduce the chances of developing autoimmune reactions since they target the immunodominant epitopes.
Delivery system and formulation with adjuvants is essential to improve immunogenicity. These methods are conjugation with carrier proteins, toll-like receptor (TLR) agonists and adsorption to nanoparticles or emulsions. In rational vaccine design, epitope mapping and bioinformatics tools come useful.
COVID-19 and New Pathogens
Peptide-based technologies found application during the COVID-19 pandemic, in form of a standalone vaccine as well as in subunit vaccines. The peptides conveying epitopes on the SARS-CoV-2 spike protein were investigated to trigger the immune effectors (neutralizing antibodies) and T-cell responses.
In addition to COVID-19, peptide vaccines are in development against influenza, Zika, dengue, HIV and hepatitis C. They are modular and can thus quickly be adapted to new variants meaning that in pandemic preparedness they have been helpful tools.
Individualised Peptide Vaccines
New directions in the field of immunotherapy of cancer have also welcomed individual neoantigen peptide vaccines which are mutated neoantigenic peptides that are specific to the individual tumors. The specialty of these vaccines consists in agreement with genomic sequencing of tumor samples and intends to invoke cytotoxic T lymphocytes focused on the tumor of the patient.
The early clinical evidence indicates that neoantigen vaccine can evoke great immune response and is highly tolerable. When combined with checkpoint inhibitors, a combination may increase the therapeutic power. Nevertheless, there exist barriers pertaining to locating the immunogenic neoantigens, and the protocols of timely developing the vaccines.
Conclusion
Innovation Peptide-based therapeutics and vaccines are an active and growing area of biomedical research. Their inherent strengths, alongside continuing improvements to the delivery platform and molecular design, have seen the generation of some highly targeted and effective therapeutic interventions in an increasingly wide range of disease areas. However, stability, delivery, and immunogenicity issues need this innovation.
New delivery techniques, including nanocarrier systems, hydrogels, and microneedle formats, and novel therapeutic constructs (such as PDCs and neoantigen vaccines) are expanding the range of uses with which peptides may be applied. With an enhanced insight into the biology of diseases and development of precision medicine, peptide-based approaches will become central in determining the future of therapeutic and preventive medicine.
It is necessary that Interdisciplinary cooperation and intense clinical assessment be continuous in order to explore the full potential of peptide-based modalities and accurately quantify patient benefit potentialities.











