Pharmaceutical 3D Printing
Small Batches Making Big Waves
Anna Worsley, CEO, FABRX-AI
Alvaro Goyanes, Co-Founder, Director, FABRX
Khalid Garba Mohammed, Senior Formulation Scientist, FABRX
Pharmaceutical 3D printing in hospitals and pharmacies is revolutionizing medicine manufacturing. Pioneering pharmacists in hospitals have started 3D printing medicine on-site to treat their patients, using pharmaceutical 3D printers designed for precision medicine. This innovative technique enhances patient clinical outcomes, reduces waste, and optimizes the supply chain, disrupting healthcare with a rapidly evolving industry.

The concept of pharmaceutical 3D printing is not new, with the phenomenon gripping the attention of the scientific community for the past years. However, only recently has this exciting new technology started to reach patients. In the 2010s, its potential in medicine mass manufacturing for complex release profiles had been taking centre stage, with real-world translation being led by companies including Aprecia Pharmaceuticals. More recently, the field has been turning to the clinical implementation of smaller scale pharmaceutical 3D printing in hospital and community pharmacy settings to benefit patients in what is commonly referred to as on-demand or point-of-care manufacturing. In this article, we discuss the progress of small-batch pharmaceutical 3D printing as a point-of-care manufacturing platform, its potential in various use cases and highlights on the evolving position of regulatory agencies to support the innovation.
Why Pharmaceutical 3D Printing is Critical for Point-of-Care Manufacturing
It is evident that one size does not fit all when it comes to drug dosing. 40-70% of off-the-shelf medicine are not effective for patients and 7% of UK hospital admissions result from related adverse drug reactions. Medical organizations and regulatory bodies around the world are calling for personalised medicine, and for children, age-appropriate medications fit-for-purpose. In fact, studies show that only 48% to 54% of all approved medicines are commercially available for pediatrics, leading to 50% of pediatric patients receiving an unlicensed or off-label prescription. The common practice to prepare these non-commercially available prescriptions is to manually prepare them via compounding. Also used for medicine shortages, compounding involves the breaking of commercially available tablets, hand-filling capsules with powders or manually measuring out liquids. This practice is associated with a myriad of problems such as over or under-dosing, instability of liquid medications, arduous frequent refill processes for long-term treatment and pour patient treatment adherence. In fact, it has been reported that 35% of pediatric non-adherence to compounded treatment is directly caused by these old-fashioned techniques.
It is easy to see how small batch pharmaceutical 3D printing can address these pressing medication problems, automating the compounding process to prepare easy-to-take exact dosages. This automation increases accessibility to truly personalized medicine on a wider scale by reducing issues surrounding compounding. Namely, by reducing risk for the pharmacist and patient while improving patient acceptability and adherence to their medicines.
Pharmaceutical 3D Printing at the Point-of-Care in Action
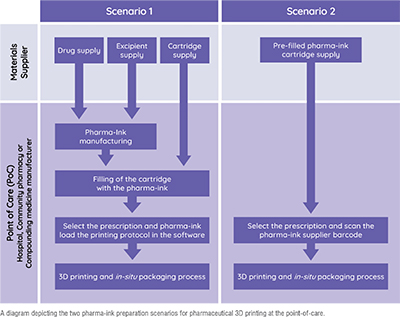
There are two scenarios for pharmaceutical 3D printing at the point-of-care, focusing on where the pharma-ink (the mixture of drug and excipients used to print medicines) is prepared. Scenario 1 is similar to standard pharmaceutical compounding workflows, preparing the drug loaded pharma-ink in the compounding pharmacy itself. Scenario 2 is when the pharma-ink preparation is outsourced, being supplied to pharmacies as pre-filled cartridges containing the drug-loaded pharma-ink, manufactured by pharmaceutical companies and CDMOs.
Scenario 1 is the easier approach to implement clinically, already being used in European compounding pharmacies for patients as standard care. Pharmacists follow normal compounding workflows and regulation, preparing the pharma-ink from scratch in the compounding pharmacy using raw drug and excipient materials. The pharmacist then fills the printing cartridge themselves, inserts it into the printer, and selects the prescription and pharma-ink protocol being used in the software and prints. Prior to use as standard care under compounding regulation, many pharmacies opt for clinical studies to be able to publish their formulation development journey and retain a research active output. These range from small paediatric studies in Spain for rare diseases to a breast cancer multi-drug polypill study involving over 200 patients in Europe’s top oncology hospital, Gustave Roussy Institute in France. Those already published have reported successful pharmacy implementation, positive staff engagement and improved patient treatment acceptability, supporting the benefits pharmaceutical 3D printing companies advertise.
Scenario 2 offers many benefits over Scenario 1, going a step further with reducing pharmacist workload, pharmacist drug exposure risk and dosing inaccuracies. Essentially the pharmacist would only need to scan the cartridge that contains the drug loaded pharma-ink, insert the cartridge into the printer, input the prescription requirements and press print. All of the pharma-ink specific activities would be outsourced or automated further. Although futuristic right now, these workflows are being investigated by pharmaceutical companies around the world, working with pharmaceutical 3D printing companies to smooth out the clinical translation process. One such project was published recently, with pharmaceutical company Losan Pharma working with pharmaceutical 3D printing company FABRX, University College London and the University of Santiago de Compostela. The team simulated scenario 2 successfully with Losan Pharma’s pre-prepared Efavirenz-loaded granulates, providing a vision of our not-too-distant future.
Additional use cases
Veterinary Medicine
Pharmaceutical 3D printing enables customized drug formulations tailored to specific animals' needs. Reflecting human medicine, it allows precise dosing, unique shapes, and flavours particular to specific animals to enhance treatment compliance; everyone with a pet knows how difficult this can be. Pharmaceutical 3D printing improves treatment effectiveness and accessibility by producing on-demand, patient-specific medications, revolutionizing veterinary care with personalized therapies for various species and animal sizes.
Astropharmacy
Space...the final frontier, and another future use case for pharmaceutical 3D printing. As far off as it sounds, more stakeholders engage in this discussion every year. NASA’s Chief Medical Officer Dr J.D Polk and Dr. Neal Zapp, the Manager of NASA’s Health and Medical Technical Authority only recently visited the world leading University College London’s Pharmaceutical 3D printing research group, The Basit Group, earlier this year. Steps to make this a reality are in discussion in order to reap the benefits on offer in time for incoming missions, for example reduced supply chain dependency and rapid, dynamic pharmaceutical preparation for the hard-to-predict future needs of astronauts on long-haul missions.
Clinical Trial batch manufacture
Pharmaceutical 3D printing has great potential for clinical and pre-clinical trials. It allows fast production of small batches containing personalized drug loading. This speeds up the process of testing multiple drug formulations and dosages while only using small quantities of experimental drug, leading to cost savings and reduced waste. There are also opportunities to bring clinical trial batch manufacture ‘closer-to-the-patient’, improving access for participants, making recruitment easier and therefore increasing representation of diverse populations. Large, international pharmaceutical companies have already started to get involved, working with pharmaceutical 3D printing experts to help implement 3D printing into their complex clinical trial workflows.
Regulatory Compliance and Standardization
Pharmaceutical 3D printing at the point-of-care can already be used as a compounding technique for personalised, non-commercially available prescriptions. Compounding regulation is how pharmacies in Europe have already started printing dosage forms for standard practice in scenario 1. However, the novelty of this innovative technology is restricting the implementation of scenario 2 where the pharma-ink is outsourced and not prepared at the point-of-care. Furthermore, there must be clear guidelines and standards to encourage pharmacists and ensure high quality of produced dosage forms. Recently, leading stakeholders in the field set-up the non-profit The International Pharmaceutical 3D Printing Initiative to help solve these issues, alongside encouraging field growth and collaboration. The initiative includes people from across the healthcare sector, including academia, hospitals, industry and regulatory bodies.
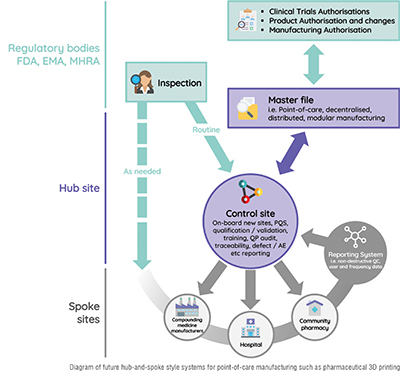
As the field grows, additional legislation will be needed to regulate the large number of printing sites popping up globally. Key regulatory authorities have already started preparing for this, with new legislation expected in the next few years from the FDA as well as the EMA in the EU, and MHRA in the UK, all with similar proposals. This point-of-care manufacturing legislation describes hub-and-spoke style systems. One hub site will regulate multiple spokes (3D printing pharmacies and hospitals) in a geographical region or commercial network and the governing regulatory body will regulate the hubs, reducing their workload. Some companies have already started to prepare for this. In fact, FABRX has built software directly mirroring this new legislation. Sharing the same name, their M3DIMAKER Studio Hub & Spoke software acts as hub and spoke access points, automatically saving every printing session at spoke sites and allowing for remote auditing for hubs.
Closing remarks
Small batch pharmaceutical 3D printing is a rapidly developing field with many exciting collaborations and projects starting in the past few years. With international pharmaceutical companies, key regulatory bodies and world-renowned research organisations getting involved, major advancements are being achieved. American organizations are beginning to catch up to the fast-moving European players who are already integrating pharmaceutical 3D printing into standard patient care. However, it is crucial that American stakeholders act swiftly to stay competitive in this rapidly evolving sector. There are ample opportunities to be had, in regular compounding workflows able to implement immediately and in the future, truly personalised medicine as part of advanced healthcare offerings. To get involved, it is advisable to reach out to stakeholders who are already active in the field. These include members of The International Pharmaceutical 3D printing Initiative (www.pharma3dpi.org) and pharmaceutical 3D printing companies. Collaborative efforts can offer valuable insights and guidance for practical implementation.
References
1. Rodríguez-Pombo, L., et al., Paediatric clinical study of 3D printed personalised medicines for rare metabolic disorders. International Journal of Pharmaceutics, 2024. 657: p. 124140.
2. Krueger, L., et al., Clinical translation of 3D printed pharmaceuticals. Nature Reviews Bioengineering, 2024.
3. Seoane-Viaño, I., et al., Translating 3D printed pharmaceuticals: From hype to real-world clinical applications. Adv Drug Deliv Rev, 2021. 174: p. 553-575.
4. The Pharmaceutical Journal (2022). 3D printing of pharmaceuticals and the role of pharmacy. https://pharmaceutical-journal.com/article/research/3d-printing-of-pharmaceuticals-and-the-role-of-pharmacy accessed 16th August 2024
5. Selmin, F., et al., Alternatives When an Authorized Medicinal Product is Not Available. Medicine Access @ Point of Care, 2017. 1: p. maapoc.0000005.
6. Goyanes, A., et al., Automated therapy preparation of isoleucine formulations using 3D printing for the treatment of MSUD: First single-centre, prospective, crossover study in patients. Int J Pharm, 2019. 567: p. 118497.
7. Denis, L., et al., Developing an innovative 3D printing platform for production of personalised medicines in a hospital for the OPERA clinical trial. Int J Pharm, 2024. 661: p. 124306.
8. Sjöholm, E., et al., Compounding Tailored Veterinary Chewable Tablets Close to the Point-of-Care by Means of 3D Printing. Pharmaceutics, 2022. 14(7): p. 1339.
9. FABRX (2024). NASA visits FABRX and the UCL 3D printing research group. https://www.linkedin.com/posts/fabrx-ltd-_spacemedicine-3dprinting-fabrx-activity-7221148381224603650-63GY/?utm_source=share&utm_medium=member_android Accessed 16th August 2024
10. Mamo, H.B., M. Adamiak, and A. Kunwar, 3D printed biomedical devices and their applications: A review on state-of-the-art technologies, existing challenges, and future perspectives. Journal of the Mechanical Behavior of Biomedical Materials, 2023. 143: p. 105930.
11. Kardas P et al., Adherence to treatment in paediatric patients – results of the nationwide survey in Poland; BMC Pediatr. 2021; 21: 16. doi: 10.1186/s12887-020-02477-z
12. Seoane-Viaño et al., A case study on decentralized manufacturing of 3D printed medicines. International Journal of Pharmaceutics: X, 2023, 5:100184
13. Monique Al, Solange Levison, Wolfgang E Berdel, Ditte Zerlang Andersen, For the Decentralised Clinical Trials Task Force, Decentralised elements in clinical trials: recommendations from the European Medicines Regulatory Network, The Lancet, Volume 401, issue 10385, April 22 2023, Page 1339, https://doi.org/10.1016/S0140-6736(23)00463-4
14. MHRA (2023). Consultation on Point of Care manufacturing. https://www.gov.uk/government/consultations/point-of-care-consultation/consultation-on-point-of-care-manufacturing Accessed 12th August 2024
15. MHRA (2023). Government response to consultation on proposals to support the regulation of medicines manufactured at the Point of Care. https://www.gov.uk/government/consultations/point-of-care-consultation/outcome/government-response-to-consultation-on-proposals-to-support-the-regulation-of-medicines-manufactured-at-the-point-of-care Accessed 12th August 2024
16. FDA (2022). Workshop on the Regulatory Framework for Distributed and Point of Care Pharmaceutical Manufacturing: An Opportunity for DM/POC Stakeholder Engagement https://www.fda.gov/drugs/news-events-human-drugs/fdapqri-workshop-regulatory-framework-distributed-and-point-care-pharmaceutical-manufacturing Accessed 16th August 2024









