Serialization and Track-and-Trace
Securing the Pharmaceutical Supply Chain
Harry Callum, Editorial Team, Pharma Focus America
Pharmaceutical Supply chain security is a necessity of serialization and track and trace technologies. These systems allow product authentication, real-time tracking, and compliance with regulation, thus helps avoid counterfeited medicines and enhance patient safety. The article examines the regulatory picture across the world, most important technologies, application issues that may arise in the implementation process, and prospective trends that facilitate the transformation toward more transparent, digitally powered pharma supply chains.

Counterfeit medicines, diversion of drugs, and illegal distribution are among the causes of mounting pressure of pharmaceutical supply chains. To the view, the safety of patients and integrity of products is central to the thinking and securing the supply chain has been at the top best priority manufacturers, regulators, and healthcare providers. Serialization and track-and-trace are the two important processes that help in determining the originality of the medicines and monitoring the control along with every phase of the supply chain.
This article describes the functionality of serialization and track-and-trace, its advantages, current regulatory framework, enabling technologies and the issues companies have in implementing them.
Understanding Serialization in Pharmaceutical Manufacturing
Serialization is the property of defining each unit of a medicinal product that can be sold with a distinctive serial number. Collectively with other identifiers, such as Global Trade Item Number (GTIN), batch or lot number and expiry date, this number will produce a unique identity per package.
The typical implementation of these codes is packaging them through high-speed printers and inspection of these codes is done through the use of vision inspector systems. It is to allow checking authenticity and tracking of the product during its path of production to dispensing.
The Role of Track-and-Trace Technologies
Whereas serialization provides the identification of individual units, track-and-trace keep tracks on the flow of these units throughout the supply chain. This process gives complete view of product, change in location and ownership processing.
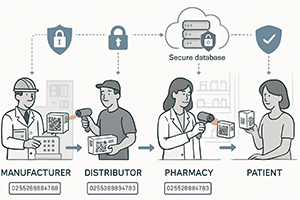
The usual technologies involve use of 2D barcodes (e.g. Data Matrix), radio frequency identification (RFID) and software tools in data capture and analysis. These schemes assist in guaranteeing that the medicines that get to the patients are genuine, intact and have been supplied through the proper channels.
Regulatory Landscape: Global Developments
Regulatory authorities and governments, the world over, have come up with systems which are applied to enforce serialization and traceability. These frameworks are meant to enhance drug safety, eliminate falsification and build a better trust on pharmaceutical products in the minds of the people.
Key Regulations Around the World
| Region/Country | Regulation | Main Requirement |
| United States | Drug Supply Chain Security Act | Full serialisation and interoperability by 2024 |
| European Union | Falsified Medicines Directive | Unique identifiers and tamper-evident packaging |
| India | Export Guidelines under D&C Act | Barcoding at secondary and tertiary packaging levels |
| Brazil | National Medicine Control System (SNCM) | Traceability through unique codes and centralised reporting |
| China | Drug Administration Law (DAL) | Traceability and data-sharing for quality assurance |
Regulatory policies are regionally different with respect to scope and technical standards and so also to timelines. International producers should harmonize with several regulations at the same time, making the task more complicated.
Strategic Value of Serialization and Track-and-Trace
Along with regulatory compliance, long-term operational and strategic advantages apply to serialisation and tracking systems.
Benefits Table
| Benefit | Description |
| Counterfeit Detection | Assists in avoiding entry of falsified products in the chain |
| Product Recall Efficiency | Facilitates the accurate identification and searching off infected batches |
| Improved Patient Safety | These guarantees medicine authenticity and traceability in points of dispense |
| Better Supply Chain | This improves demand planning, tracking of shipments, and inventory management |
| Regulatory Readiness | Enables adherence to diverse global provisions of traceability |
Such advantages help to enhance risk management and the ability of business to be resilient.
Technologies Supporting Implementation
Achievement of successful implementation is dictated by softwares and hardware integration in packaging systems, warehousing, and logistics.


• Packaging Line Equipment: Printers, scanners and checkers in order to apply and check codes properly
• Software Platforms: Services coordinating data gathering, reporting, as well as exchange at various levels (line packaging to cloud)
• Data Integration: Integration with ERP, warehouses and supply chains to allow smooth tracking
• Cybersecurity: Security of serialisation information and prohibition of misuse or unlicensed accesses
High technology including cloud computing and machine learning are becoming more and more utilized to handle big data and improve decision-making.
Serialization Across Packaging Levels
Serialisation usually starts at the unit-level packaging (also referred to as primary packaging) to achieve full trace of the distributed product, but commonly continues either to secondary and tertiary level packaging.
• Example of primary packaging are vials, blister packs, or bottles. They may not in all cases be serialized, but they are correlated with outer package codes by aggregation.
• The most popular level of the 2D barcodes or serial codes applies to secondary packaging, like a carton or a box. It is the central source of product verification by the pharmacists and distributors.
• Shipping cartons or pallets are covered by the term tertiary packaging. They are normally marked with summary codes which ties them all back to all the items within it enhancing efficiency in the warehouse and in logistics.
Best practices of aggregation, the practice of connecting individual units to cases and pallet, increasingly emerge in non-requiring environments. It eliminates the need to scan manually, accelerates processing in operating distribution centres, eases the process of verification during customs or border operations.
Stakeholder Collaboration is Key
Effective serialization and traceability implementation would be successful when it involves a very close interaction between various stakeholders, such as:
• Pharmaceutical manufacturers are companies which carry out marking with serial numbers as well as ensure compliance
• The machinery suppliers of packaging who have to modify systems to accommodate high-speed, high-accuracy coding and inspection
• The software vendors - those that provide traceability systems using wider enterprise platforms
• Wholesalers and distributors, who should be capable of reading and verifying serial number when the and product enters the downstream chain
• Verification and authentication can be at point of dispensing in pharmacies and hospitals
All these stakeholders will have to play on the same level of systems and exchange information in a secure and sufficient way. In most nations, the regulators have central databases upon which the serial numbers are posted and checked. The Interoperability and standardisation are very important in the effectiveness of these systems across regions.
Real-World Applications and Industry Response
Serialisation programmes at global facilities have been adopted by different pharmaceutical firms and contractors. Such programmes are usually piloted on a few lines of packaging and are extended across the lines.
In practice implementation schedules are related to a number of factors:
• The degree of the current automation in packaging activities
• Digital-infrastructure maturity (e.g. data systems, ERP integration)
• The volume and intricacy of product lines and manufacturing.
• Deadlines of particular country regulations (e.g. DSCSA 2024 in the U.S.)
Serialisation has already started to be invested in by the donor-funded programs and global health organisations in some of the low- and middle-income countries to provide smooth supply of the essential medicines. This trend emphasizes the increased focus of the global community on the necessity to have pharmaceutical products that can be tracked and which have certain properties that may not be controlled.
Expanding Role in Patient Engagement
Although regulatory and operational requirements are the initial aims of serialization, it can also assist in patient-centric activities. For example:
• Product Scannable QR codes on the packaging can serve as the source of product information, instruction of use or even as the substitute to digital leaflets.
• Serial numbers can be used by the patient to prove authenticity via websites or applications.
• In the future, it may be supported by automotive monitoring through integration with electronic health records (EHRs).
With increases in demand among patients regarding visibility and digital connection, these attributes can be expected to turn into more of a standard practice when it comes to designing pharmaceutical packages.
Challenges in Execution
Although they have multiple benefits, serialisation and traceability systems implementation create some challenges to pharmaceutical manufacturers:
• Cost and Resource Requirements: This may be a high-cost project regarding equipment, software, and talented people in the beginning
• System Compatibility: If the Legacy systems are not upgraded, new traceability functions might not be supported by legacy systems
• Coordination on the Global Scale: Integrating the operation with various global rules is a resource consuming process.
• Training and Change Management: The staff members require substantial training to deal with new work practices and technologies
When dealing with these issues, there is a probable need to coordinate functions in technical, regulatory and supply chain areas.
Future Outlook: Towards a Digitally Enabled Supply Chain

Digitisation of the pharmaceutical industry is leading serialisation to increasingly become a constituent of a larger strategy to create fully connected and intelligent supply chains.
New developments are:
• Predictive Analytics: To predict how the supply chain may be disrupted or have changes in demand.
• Integration of blockchain: provision of secure decentralised platforms to authenticate the product and transfer data
• Smart Packaging: Sensors and connectivity can make it possible so that Smart Packaging can be used to monitor conditions, track expiry, or automate replenishment.
Such technologies can transform the supply chain to be proactive as opposed to reactive which will enhance delivery of products as well as offering of services.
Conclusion
Track-and-trace systems and Serialization are the parts of pharmaceutical supply chain which are crucial to the security of the chain. They facilitate regulatory address, aid in countering counterfeit medicines and increase manufacturing to dispensing transparency. The option might not be easy to roll out, and the scheme could be challenging and costly, but the end result on the patient safety, efficiency of operations, and trust by the global community would be notable.
Since the technology is changing and as regulatory needs are becoming increasingly demanding, pharmaceutical companies can take this chance and incorporate these systems into wider digital plans, thereby making medicines and supply chains much safer everywhere in the world.











