AI in R&D Platforms
Platform or Perish The Data Infrastructure Divide
Lavinia Woodward, Senior Product Developer, Beacon
Despite AI investment in pharma being predicted to reach $16.5 billion by 2034, 75% of companies continue to achieve minimal returns due to fragmented implementations. Success requires unified platform architecture, not isolated AI applications. Industry leaders demonstrate two proven pathways: Novartis's consolidate-then-scale approach and GSK's embed-from-day-one strategy. Both eliminate data silos and enable enterprise-wide AI deployment. Companies must urgently choose their transformation path; those maintaining fragmented systems risk competitive extinction as platform-native competitors cut development timelines by 50% through superior R&D data infrastructure.

Pharma’s AI Tipping-Point
By 2034, investment in AI for pharmaceutical applications will increase from $1.9 billion to $16.5 billion, yet three-quarters of companies continue to experience minimal returns. McKinsey estimates the potential value of generative approaches at $60–110 billion annually, while analysts project that AI will drive half of FDA approvals by 2030.
Recursion Pharmaceutical’s Experimentation OS (Recursion OS) exemplifies the transformative power of AI-embedded platforms. Through seamless integration of automated wet-lab robotics, phenomics, and machine learning within a unified architecture, target-to-IND took just 18 months; the industry standard is 42-months. In this era of unprecedented AI transformation, ‘fortune favours should be brave; ’ only organisations that commit fully to AI-native platforms will capture the true value of R&D transformation. This article provides mid- and large-cap biopharma incumbents with the strategic playbooks to demonstrate such courage, offering proven frameworks to scale AI across their R&D platforms.
The core challenge lies not in AI technology itself, but in platform architecture. Most pharmaceutical companies deploy AI as isolated applications rather than integrated platform capabilities. Success requires enterprise infrastructure where AI embeds throughout workflows. Data must flow seamlessly across functions. Only then can insights compound with each experiment and market signal. The blueprint for success exists; the critical question is which organisations will take decisive action, and which will continue relying on bolt-on solutions, accepting the inevitable competitive disadvantages.
Platform Infrastructure as Competitive R&D Advantage
The Cost of Fragmentation
With hundreds of AI use cases under exploration, and each pilot project costing between US$50 000 and US$1 million, the cost of fragmentation quickly becomes prohibitive. McKinsey observes that model building accounts for just 15 % of effort; most of the effort goes into stitching algorithms to dispersed data and workflows. Without a unified platform, AI collapses into costly, isolated islands. Unsurprisingly, a Norstella survey found 42 % of pharmaceutical organisations naming data integration as the biggest obstacle to scaling AI.
The true price extends beyond capital. Discovery, research, clinical, and commercial teams each run siloed pilots, duplicating infrastructure, development, and operational costs This duplication of work has been calculated to add 30% to the spend. Yet, financial cost is the smaller loss: AI models confined to departmental silos forgo cross functional data that would enhance accuracy and force teams to rebuild similar models elsewhere. Without platform integration, breakthroughs, trial insights and market signals remain locked in their silos, stalling the compounding learning that should accelerate innovation across the enterprise.
Eliminating Data Silos Through Platform Thinking
Despite widespread digitisation, research scientists still spend 40–60% of their time manually harmonising, cleaning, and formatting data instead of generating insights. The bottleneck is not data scarcity but the absence of automation and AI powered integration across R&D systems. Every disconnected repository is a lost learning opportunity; without a robust data foundation, even state of the art models underperform. In these circumstances, a self-reinforcing learning loop - where data feeds models, insights guide experiments, and results enrich the data - never forms. Creating these loops within R&D rests on three capabilities: (1) enterprise wide access to unified datasets; (2) reusable AI services that both consume and contribute to those datasets; and (3) automated feedback capture so each result improves the next experiment.
AbbVie is leading one of the largest data convergence initiatives in the biopharma industry. Data is collected, harmonised, and made available across the entire organisation as part of a culture of sharing data across functions, ensuring everyone benefits. The AbbVie R&D Convergence Hub (ARCH) is an example not only of AI models powered by an integrated platform, but of AI/ML capabilities cleaning, processing, and harmonising data. By centralising and connecting data from more than two hundred internal and external sources, ARCH helps to build the data ecosystem through which it is embedded.
Yet building these foundational capabilities is merely table stakes. The pharmaceutical companies that transform data infrastructure into competitive advantage are those that move beyond integration to acceleration, deploying AI not just to clean data but to fundamentally reimagine how R&D platforms operate. The speed and scale at which companies execute this transformation now determines whether they lead or follow in the AI-enhanced future of drug development.
The Architecture Divide: How Lilly Widened the Chasm
Eli Lilly demonstrated meteoric ascent in CB Insights' Pharma AI-Readiness Index, from 14th to 1st between 2023 and 2025. This rise shows that with strong leadership and a clear strategy, organisations can make rapid, significant improvements to AI across the entire R&D data ecosystem, securing a decisive edge over piecemeal adopters.
Lilly’s success stemmed from three interconnected approaches. First, Lilly scaled its internal data infrastructure by expanding data science, engineering, and IT teams, thereby creating the foundational layer necessary for enterprise-wide AI deployment. Second, rather than limiting AI to specialist teams, through platform-native functionalities and training, access was democratised, enabling organisation-wide value creation. Third, Lilly orchestrated 13 AI investments in 2024 alone, outpacing every other pharmaceutical company through a portfolio approach that spanned drug discovery, medical devices, and regulatory solutions.
The generative AI for antimicrobial discovery collaboration with OpenAI exemplifies how platform architecture enables rapid capability expansion. Rather than building in isolation, Lilly leveraged its robust data infrastructure to integrate generative AI directly into existing drug discovery workflows. This ability to rapidly absorb and deploy advanced AI technologies represents the new competitive reality in pharmaceutical R&D. In May of this year, Lilly’s Executive Vice President and Chief Information and Digital Officer, Diogo Rau warned that companies and institutions that don't invest in AI now won't be around in 2050. As the chasm widens between companies that scale AI throughout their R&D platforms, and those that do not, this may come true sooner than predicted.
Destination: AI-Native R&D Platforms
Proven Blueprints for AI Transformation
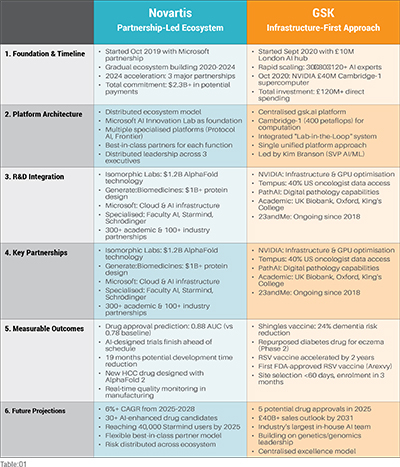
The race to build AI-native R&D platforms has produced two distinct blueprints for transformation. GSK pursued an infrastructure-first approach, investing £120M to build internal AI capabilities and secure dedicated supercomputing power. Novartis chose a partnership-led ecosystem strategy, committing $2.3B across specialised AI collaborators to access best-in-class technologies. While their paths diverge dramatically, both companies share the same destination: fully integrated AI platforms that transform how drugs are discovered and developed. This analysis examines the six critical stages where these contrasting strategies playout.
Key Strategic Differences
GSK: Concentrated investment in internal capabilities and infrastructure, emphasising control and deep integration with focus on genetic-driven discovery.
Novartis: Distributed risk and expertise across specialised partners, prioritising flexibility and comprehensive process transformation across entire R&D value chain.
Beyond Strategy: The Reality of Implementation
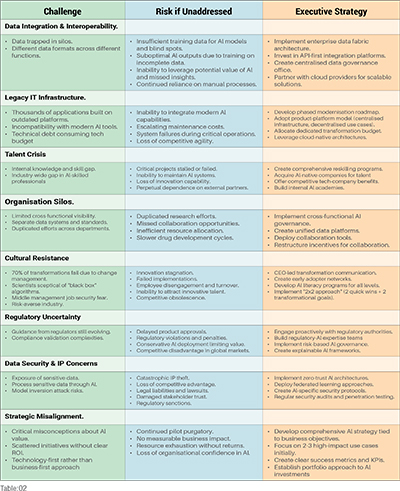
While GSK and Novartis have chosen different paths to build AI-native R&D platforms, both face the same fundamental challenge: scaling AI from promising pilots to enterprise-wide transformation. The common obstacles encountered on the journey from proof-of-concept to platform-wide integration of AI, together with proposed solutions, are outlined below.
Avoiding Common Roadblocks
While these case studies demonstrate clear routes to AI-native R&D platforms, execution remains complex. Even organisations following Novartis's consolidated approach or GSK's embedded strategy often face implementation challenges that can derail transformation efforts. Figure 1 highlights eight common roadblocks and provides executive actions to navigate each.
These roadblocks are common and can be overcome, but only with executive commitment and systematic execution. The question is not whether to transform, but how quickly you can navigate these challenges before competitors establish insurmountable platform advantages.
Conclusion
The journey from fragmented AI applications to integrated AI-powered R&D platforms represents fundamental transformation in how pharmaceutical companies create value. The path is clear: move from treating AI as point solutions to scaling AI throughout the drug development continuum. Delay now means ceding leadership to faster-moving competitors. When platform-native companies can cut development timelines in half and potentially double success rates through superior R&D infrastructure, the cost of platform fragmentation becomes unquantifiable.
The platform infrastructure exists. The integration blueprints are proven. The only question is whether pharmaceutical leaders will build unified R&D platforms underpinned by AI or watch as others define the industry's future.
References
PwC. Next in pharma 2025: The future is now. PwC. Published January 8 2025. Accessed July 5 2025. https://www.pwc.com/us/en/industries/pharma-life-sciences/pharmaceutical-industry-trends.html PwC
Precedence Research. AI in pharmaceutical market size, share, and trends 2025 2034. Precedence Research. Updated January 16 2025. Accessed July 5 2025. https://www.precedenceresearch.com/ai-in-pharmaceutical-market Precedence Research
Shah B, Viswa CA, Zurkiya D, Leydon E, Bleys J. Generative AI in the pharmaceutical industry: moving from hype to reality. McKinsey & Company. Published January 9 2024. Accessed July 5 2025. https://www.mckinsey.com/industries/life-sciences/our-insights/generative-ai-in-the-pharmaceutical-industry-moving-from-hype-to-reality McKinsey & Company
Chmielewska I, Caruso S. Assessing current AI trends in drug development: adoption and transformation (part 1). Norstella. Published October 31 2024. Accessed July 5 2025. https://www.norstella.com/assessing-current-ai-trends-drug-development-part-1/ Norstella
Recursion Pharmaceuticals Inc. Recursion announces FDA clearance of investigational new drug application for REC 1245, a potential first in class RBM39 degrader for biomarker enriched solid tumors and lymphoma. GlobeNewswire. Published October 2 2024. Accessed July 5 2025. https://www.globenewswire.com/news-release/2024/10/02/2956875/0/en/Recursion-Announces-FDA-Clearance-of-Investigational-New-Drug-Application-for-REC-1245-a-Potential-First-In-Class-RBM39-Degrader-for-Biomarker-Enriched-Solid-Tumors-and-Lymphoma.html GlobeNewswire
Foley P. $25B potential in accelerating AI’s impact and value in pharma. Pharmaceutical Executive. Published April 29 2025. Accessed July 5 2025. https://www.pharmexec.com/view/25-b-potential-accelerating-ai-impact-value Pharmaceutical Executive







