Analytical Techniques for Antibody-Drug Conjugates
Delivering Clarity and Confidence to Cancer Therapeutics
Vara Prasad Chandrabatla, Associate Director, Veranova
Antibody-drug conjugates (ADCs) enhance cancer therapy by combining monoclonal antibodies with cytotoxic drugs, targeting cancer cells while minimizing side effects. This paper reviews key analytical techniques for ADC characterization, including HPLC and mass spectrometry, and discusses innovations and challenges in ADC development and manufacturing.
Analytical Techniques for Antibody-Drug Conjugates: Delivering Clarity and Confidence to Cancer Therapeutics
Antibody-drug conjugates (ADCs) are a major advancement in cancer therapy, combining monoclonal antibodies with cytotoxic drugs to target and destroy cancer cells while sparing healthy tissues.
This enhances treatment effectiveness and reduces side effects. ADCs consist of three key components:
• Tumor-specific antibody: Targets cancer cells.
• Cytotoxic payload: Kills the cancer cells.
• Chemical linker: Attaches the drug to the antibody and controls its release.
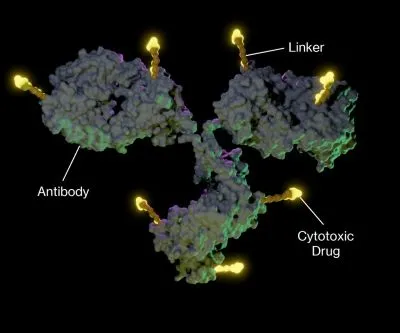
Antibody-drug conjugates (ADCs) have indeed revolutionized cancer treatment by combining the specificity of monoclonal antibodies with the potency of cytotoxic drugs. Here's a detailed overview of the current analytical landscape for ADCs:
Physicochemical Characterization
Physicochemical characterization involves analyzing the structural and chemical properties of ADCs. Techniques such as size exclusion chromatography, reversed-phase chromatography, and cation exchange chromatography are used to assess the aggregation, monomer content, and charge variants of ADCs. These methods help ensure the consistency and stability of ADCs throughout their development and production.
Biological Functionality
The biological functionality of ADCs is crucial for their effectiveness. ADCs target cancer cells with high precision, minimizing off-target effects. They can also be combined with other therapies, such as immune checkpoint inhibitors, to enhance their anti-tumor activity. Understanding the mechanisms of action and optimizing the interaction between the antibody, linker, and payload are essential for improving ADC efficacy.
Stability Testing
Stability testing is vital to ensure the safety and efficacy of ADCs over their shelf life. This involves assessing the stability of both the antibody and the conjugated drug under various conditions. Factors such as linker chemistry, conjugation sites, and drug-to-antibody ratio (DAR) distribution play significant roles in determining the stability of ADCs .
Emerging Innovations
Innovations in ADC design are continuously enhancing their therapeutic potential. Advances in antibody engineering, novel linker technologies, and next-generation cytotoxic drugs are improving the stability, specificity, and efficacy of ADCs. These innovations are paving the way for more effective and safer cancer treatments.
Obstacles in Ensuring Product Quality
Ensuring the quality of ADCs involves overcoming several challenges in their development and manufacturing. Issues such as aggregation, solubility, and thermal stability can affect the potency and shelf life of ADCs. Implementing robust analytical techniques and optimizing the production process are essential for maintaining high-quality ADCs
Chromatographic Techniques
• Size-Exclusion Chromatography (SEC): Detects aggregation or fragmentation.
• Hydrophobic Interaction Chromatography (HIC): Determines drug-to-antibody ratio (DAR).
• Reversed-Phase HPLC (RP-HPLC): Evaluates payload stability and release profile.
Mass Spectrometry (MS)
• Intact Protein MS: Measures molecular weight and DAR.
• Peptide Mapping: Identifies conjugation sites and modifications.
• Advanced Dissociation Techniques: Characterizes linker and payload attachment.
Spectroscopic Methods
• UV-Vis Spectroscopy: Determines DAR and drug loading.
• Circular Dichroism (CD): Evaluates secondary and tertiary structures.
• FTIR Spectroscopy: Analyzes functional groups and structural changes.
Functional Assays
• Binding Affinity and Specificity: Verified by SPR and ELISA.
• Cell-Based Cytotoxicity Assays: Assess ADC's ability to kill cancer cells.
Pharmacokinetic and Pharmacodynamic Evaluations
• Pharmacokinetics: Tracks distribution, metabolism, and excretion.
• Pharmacodynamics: Measures effectiveness in animal models or clinical studies.
Stability and Quality Control
• Stability Testing: Ensures ADCs withstand environmental and processing conditions.
• Quality Control: Maintains therapeutic consistency and safety.
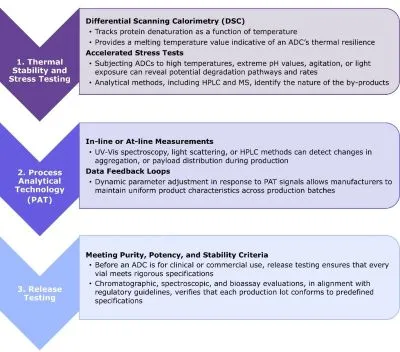
Figure 1: Stability and Quality Control Studies throughout the ADC Lifecycle
Testing at various stages of development provides knowledge of the acceptable parameter space requirements for producing an ADC with the appropriate stability and activity profile.
Emerging Innovations in ADC Analytics
Ongoing technological progress has transformed many facets of ADC analysis. Newly developed tools and approaches offer deeper insights into the behaviour of antibody-drug conjugates, enabling better control and faster development timelines.
1. Artificial Intelligence and Machine Learning
Artificial intelligence and machine learning algorithms can assimilate data from multiple analytical platforms, identifying subtle patterns and generating predictive models for critical quality attributes. By using historical and real-time data, these tools will be able to:
• Anticipate Batch Variability: Predict whether minor changes in raw materials or environmental conditions could lead to off-specification products.
• Optimize Conjugation Parameters: Suggest ideal reaction times, temperatures, or linker chemistries based on complex datasets.
• Facilitate Regulatory Compliance: Support advanced data analytics that demonstrate process understanding to regulatory bodies.
2. Multi-Omics Approaches
Multi-omics platforms, including proteomics and metabolomics, provide comprehensive snapshots of interactions between the ADC and biological systems. By correlating molecular profiles with functional outcomes, researchers can pinpoint factors contributing to resistance or off-target effects. This integrated perspective is particularly valuable in preclinical studies, where deeper mechanistic insights can inform better drug design.
3. Cutting-Edge Separation and Detection Methods
Multi-dimensional chromatography, microfluidic platforms, and high-resolution mass spectrometers are pushing the boundaries of ADC analysis. These technologies allow for:
• Higher Throughput: Rapid screening of numerous samples, facilitating parallel optimization.
• Enhanced Resolution: Better separation of different conjugation species and isoforms, enabling improved quality control and deeper structural insights.
• Miniaturized Systems: Reduced sample and reagent requirements, leading to cost savings and environmental benefits.
4. Next-Generation Bioassays
The expanding complexity of antibody-drug conjugates, with novel payloads and linkers, necessitates advanced bioassay designs:
• Multiplexed Flow Cytometry: By simultaneously measuring multiple biomarkers, researchers can rapidly assess the cellular impact of ADCs under various conditions.
• Label-Free Biosensors: The real-time detection of antibody-antigen interactions, without the need for secondary reagents, allows streamlining of functional assays.
These innovations underscore the dynamism of the antibody-drug conjugate field, where analytical sophistication grows in parallel with novel therapeutic designs.
Approaching the Principal Challenges in ADC Analytics
The possibility of precision, personalized therapy offered by ADCs has driven advancements in analytical techniques. This evolution is key to the improvements seen across generations of ADCs. Despite the visible progress, several hurdles persist in the analytical characterization and manufacture of antibody-drug conjugates (Figure 2), with opportunities for collaborative solutions.
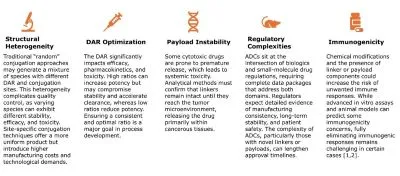
Figure 2: Challenges in Characterization and Manufacture of ADCs
These challenges necessitate ongoing collaboration among pharmaceutical companies, academic researchers, and regulatory bodies to refine both the science and the regulations governing antibody-drug conjugates.
One of the most noteworthy advancements in ADC manufacturing involves site-specific conjugation, in which the cytotoxic payload is attached to defined amino acid residues. By reducing heterogeneity, site-specific technologies can produce antibody-drug conjugates with more predictable DAR and pharmacokinetics. Analytical verification of site-specific attachment employs a combination of top-down mass spectrometry for intact protein mass analysis, and peptide mapping to confirm that conjugation occurs exactly where intended [4]. While this approach can result in superior quality and performance, it also elevates manufacturing costs and complexity, illustrating the balance developers must strike between product uniformity and affordability.
The agility and expertise of established CDMOs help developers overcome these challenges and provide the extra bandwidth and dedication that streamline drug development. The role of the CDMO evolves over time and so must the development strategy. Experience with phase-appropriate development and navigating the regulatory landscape allows the right CDMO to provide holistic support. The transition from drug discovery to market is a complex process; each phase demands a tailored approach. Early phase development serves as the proof of concept and focuses on speed, flexibility, and small-scale manufacturing. This involves selecting a target antigen and antibody that minimizes off-target toxicity and testing different linker-payload combinations and conjugation methods with the goal of a reproducible, consistent DAR. At this stage, the process may not be fully validated. Mid-phase development focuses on scalability and process refinement with more developed in-process controls. More robust analytical characterization is required and initial planning for late-stage technology transfer begins. During late phase development, ADC manufacturing must be cGMP compliant, scalable, and ready for validation. Long term stability studies and navigation of the regulatory landscape are a must at this stage. CDMOs with proven experience across the development lifecycle establish regulatory compliance procedures early on, enabling smooth, efficient preparation for late-stage submissions. Veranova’s state-of-the-art equipment provides end-to-end characterization using phase-appropriate analytical methods. Testing capabilities cover both functional and physicochemical properties. Extensive experience with linker molecules and highly potent active pharmaceutical ingredients, including cytotoxic payloads, positions Veranova as a key strategic partner for ADC development.
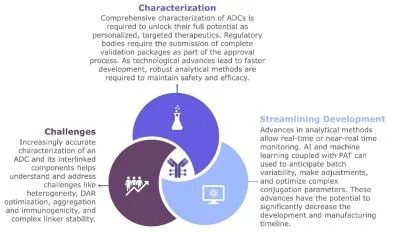
Toward the Future: Opportunities and Trends
The potential of ADCs continues to drive innovation in oncology. Ongoing developments point to an even more sophisticated future for ADCs and the analytics that support them.
1. Automation and Robotics
Automated platforms for sample handling, chromatography, and mass spectrometry enhance reproducibility and speed, reducing errors and freeing scientists for data interpretation
2. Personalized Medicine Approaches
ADCs may be tailored to individual patient profiles, requiring agile analytical methods adaptable to unique samples or small production batches
3. Regulatory Harmonization
Global regulatory agencies are working towards harmonized guidelines to streamline approval processes and accelerate access to innovative treatments
4. Integration of Artificial Intelligence
AI techniques like predictive modeling and automated anomaly detection can reduce drug development timelines while maintaining high quality standards. [3].
5. Expansion into Non-Oncology Indications
Researchers are exploring ADCs for autoimmune diseases, infectious diseases, and other conditions, necessitating distinct analytical endpoints and regulatory pathways
Conclusion
ADCs combine the targeting capability of antibodies with the cytotoxic effect of small-molecule drugs, offering effective tumor treatment with reduced systemic toxicity. Robust analytical techniques are essential throughout ADC development, from prototype screening to full-scale manufacturing and quality control. Innovations in AI, multi-omics, and advanced separation technologies will help overcome existing challenges, ensuring ADCs remain a cornerstone in modern oncology and beyond
References
[1] Fahim M, Emer E, Connor A, Gallo J. Combined approaches to reduce immunogenicity of protein therapeutics. Ther Deliv. 2013;4(5):659-72.
[2] Lambert JM, Chari RV. Ado-trastuzumab Emtansine (T-DM1): An armed antibody to combat HER2-positive breast cancer. CA Cancer J Clin. 2014;64(6):457-67.
[3] Chen H, Lin Z, Arnst KE, Miller DD, Li W. Tubulin inhibitor-based antibody drug conjugates for cancer therapy. Molecules. 2017;22(8):1281.
[4] Beck A, Goetsch L, Dumontet C, Corvaia N. Strategies and challenges for the next generation of antibody drug conjugates. Nat Rev Drug Discov. 2017;16(5):315-337.
[5] Alley SC, Benjamin DR, Jeffrey SC, Okeley NM, Meyer DL, Sanderson RJ, et al. Contribution of antibody binding affinity to the anti-tumour efficacy of antibody drug conjugates. Bioconjug Chem. 2008;19(3):759-65.










