Long-acting Drug Delivery Systems
Present and Future
Prof. Eneko Larraneta, Chair in Pharmaceutical Materials Science, School of Pharmacy, Queen’s University Belfast
Dr. Juan Domínguez-Robles, Ramon y Cajal Fellow, Departamento de farmacia y tecnología farmacéutica, Universidad de Sevilla
Conventional drug delivery methods face challenges like fluctuating drug levels and poor bioavailability. Long-acting drug delivery systems (LADDS) provide steady drug release, improving efficacy and safety, especially for chronic conditions. Innovations in materials, nanotechnology, and 3D printing are driving LADDS development, with the market valued at $11.41 billion in 2022.
Current clinical therapies rely predominantly on intermittent drug administration, with oral and intravenous routes being the most employed. These routes deliver high concentrations of drugs into the bloodstream shortly after administration. However, drug levels often fall below therapeutic thresholds within a few hours, leading to the so-called “peak-and-valley” effect. This fluctuation is far from ideal, as excessively high drug concentrations can be toxic, while insufficient levels fail to provide therapeutic benefit.
The oral route poses additional challenges. Drugs administered orally must withstand the harsh conditions of the gastrointestinal tract, including an acidic environment and the presence of numerous enzymes that may degrade the drug before absorption. Furthermore, first-pass metabolism in the liver significantly reduces the bioavailability of many therapeutic compounds. To address these limitations, multiple doses are often required to maintain drug concentrations within the therapeutic window, which is crucial for managing chronic and long-term conditions. This is a pressing issue, as the incidence of chronic conditions is rising globally due to the ageing population.

Some medical conditions demand high drug concentrations at specific sites of action. While increasing the dose can achieve this, it may result in toxic levels in the bloodstream or other organs. Consequently, there is a pressing need for advanced drug delivery systems capable of providing long-term drug delivery. These systems aim to keep drug concentrations within the therapeutic range, thereby maximising efficacy while minimising toxicity and side effects. Long-acting drug delivery systems (LADDSs) represent a promising solution to these challenges.
LADDSs include a wide range of designs, including implants, nanoparticle-based formulations or in situ forming gels among others. The development of these systems requires a multidisciplinary approach, integrating material science, engineering, pharmaceutical sciences, biology, and medicine. The origins of LADDS date back to the 1930s, when hormone-loaded pellets were implanted subcutaneously in livestock to enhance growth efficiency, revolutionising meat production. This technology was later adapted for medical use, such as treating premature menopause in women.
Although LADDS were first introduced nearly 90 years ago, interest in these systems has grown significantly over the past two decades (Figure 1). Their potential extends beyond academic research, as pharmaceutical companies actively invest in developing new LADDS products. These advancements are often part of lifecycle management strategies for existing drugs, as well as for new therapeutic compounds. The global long-acting drug delivery technologies market was valued at $11.41 billion in 2022 and is projected to grow at a compound annual rate of 11.31% through to 2033. This growth reflects the increasing recognition of LADDS as a viable alternative to conventional drug delivery methods.
Recent advances in manufacturing techniques and micro/nanotechnology have expanded the capabilities of LADDS, enabling the creation of more sophisticated systems with enhanced properties tailored to specific clinical needs. For example, nanoparticle-based suspensions and biodegradable implants have emerged as effective strategies to achieve sustained drug release.
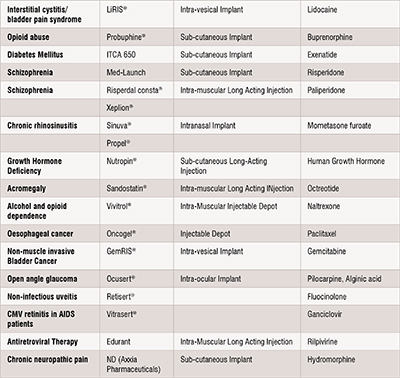
LADDS have been utilised across a broad range of medical applications, including contraception, cancer treatment, ocular conditions, pain management, nasal polyps, and disorders affecting the central nervous system. These systems employ diverse materials and methodologies in their design and fabrication. LADDS can take various forms, including solid implants, depot-forming implants, and long-acting injectable formulations. Table 1 shows a summary of commercial LADDS products. A thorough list of approved and discontinued products can be found here:
A wide array of materials is used to create these systems, ranging from metals and biodegradable polymers to non-biodegradable polymers, hydrogels, and self-assembled gels. Each material type contributes unique properties, enabling tailored designs to meet specific therapeutic needs.
As previously mentioned, the development of LADDS requires a multidisciplinary approach. Chemistry plays a key role in many of the latest advancements in this field. Researchers are continually synthesising new polymers and drug complexes to achieve sustained drug release. These polymers can be used to create nanoparticles for drug encapsulation, in situ forming implants, or pre-formed solid implants. One of the most common strategies involves the development of thermoresponsive gels, which remain liquid at room temperature but form gels at physiological temperatures. These gels, often composed of biocompatible polymers such as lactic/glycolic acid polymers and poly (ethylene glycol), can trap drugs within their structure, enabling their gradual release.
Another approach is to covalently bind drug molecules to polymers or other compounds via ester bonds, allowing controlled drug release through hydrolysis. This strategy has been employed in approved prodrugs such as paliperidone palmitate. Additionally, drugs can be combined with polymers to create "drugamers," which depend on enzymatic cleavage to release the active compound from the polymer backbone. Such formulations can be administered via conventional injection. More complex systems have also been developed, such as phosphorylated peptide-based injectable gels. In these systems, drugs are linked to peptide chains, which undergo dephosphorylation post-injection, leading to in situ gel formation and subsequent drug release via hydrolysis. However, the creation of new chemical entities poses regulatory challenges for market approval. An alternative is to chemically combine drugs with already approved polymers or molecules, facilitating a smoother path to clinical translation.

Beyond chemical modifications, alternative approaches exist to control drug delivery. Engineers have extensively developed devices capable of sustaining drug release over prolonged periods. A straightforward method involves embedding drugs within biocompatible polymers to create matrix-type implants, often manufactured via hot-melt extrusion. Process optimisation is crucial in achieving the desired drug release rate, as factors such as drug crystallinity and polymer-drug interactions significantly impact release kinetics. However, this optimisation process can be complex and typically requires substantial amounts of drug and polymer materials. Recently, Vacuum Compression Moulding has emerged as a promising technique for formulation screening using minimal material quantities, potentially accelerating the development of hot-melt extrusion and injection moulding-based formulations.
In addition to matrix-type implants, engineers have developed more sophisticated systems in which drugs are encapsulated within semipermeable membranes to regulate release (31). By controlling membrane porosity—ranging from nanometres to microns—the permeability of drugs can be finely tuned. Compared to matrix-type implants, reservoir implants offer a key advantage: they can achieve zero-order release kinetics, preventing initial burst release. Furthermore, biodegradable polymers can be used for membrane fabrication, eliminating the need for implant removal. Additionally, these systems can be prepared to be refilled externally after depleting their cargo.
Pharmaceutical formulation is another crucial aspect of LADDS development. The combination of drugs with appropriate excipients or polymers offers a relatively simple and cost-effective means of enhancing drug delivery. Unlike chemical synthesis, which involves the creation of new chemical entities, using approved excipients can expedite clinical translation. This approach is applicable across various LADDS technologies, including micro- and nanoparticle-based formulations that encapsulate drugs within pharmaceutical-grade polymers. Another well-established strategy involves combining drugs with thermoresponsive polymers such as Poloxamers, along with excipients that modify gel formation and release profiles.
Formulation strategies are also essential for solid implantable devices. Selecting the right excipients for incorporation into polymers during hot-melt extrusion can significantly alter drug release from matrix-type implants, making excipient choice critical in the design of these delivery systems. Additionally, complex reservoir-type implants rely on drug solubility to achieve optimal release. Again, the choice of excipients in the implant core can influence device performance. For example, cyclodextrins have been used in core-shell implants to enhance the release of hydrophobic drugs. Alternatively, biodegradable polymers capable of generating a micro-acidic environment upon degradation can be employed to improve the solubility of certain hydrophobic drugs.
Through these multidisciplinary approaches—spanning chemistry, engineering, and pharmaceutical formulation—LADDS continues to evolve, offering innovative solutions for sustained drug delivery. While LADDS offer numerous advantages over conventional drug administration methods, they are continually evolving to overcome existing limitations. Traditional LADDS, such as solid implants, nanoparticulate suspensions, and injectable formulations, provide sustained drug release but often require invasive procedures for administration. Implantable LADDS necessitate the use of a trocar or minor surgery, while injectable formulations, though less invasive, can still pose challenges, such as the generation of medical waste and the risk of needlestick injuries. Additionally, both methods require healthcare professionals for administration, increasing treatment costs.
To address these challenges, researchers have been developing minimally invasive alternatives. One promising approach involves the use of microarray patches (MAP) to administer nano/microparticulate-based LADDS. These devices feature arrays of tiny projections that penetrate the skin’s outer layer painlessly, creating microchannels for drug delivery. When combined with water-soluble polymers, MAP dissolve upon insertion, releasing the long-acting formulation into the skin, where the drug is gradually absorbed. Importantly, MAP can be self-administered, reducing reliance on healthcare professionals and improving patient accessibility. Although they may not deliver the same volume of medication as conventional injections, their ease of use and painless application make them a viable alternative for certain therapies, such as long-acting anti-HIV formulations. Furthermore, hybrid MAP with implantable PLGA tips, which remain within the skin as microimplants after patch removal has been recently developed. This system enables sustained intradermal drug release while ensuring easy and complete removal of the hydrogel base.
Another innovative development is the use of biodegradable microimplants, which employ longer, individual needles made from degradable polymers. These devices function similarly to MAP but are designed for the delivery of biopharmaceuticals, providing controlled drug release over time. Additionally, novel LADDS approaches seek to improve drug release precision. Traditional systems rely on passive diffusion or slow solubilisation, but advanced implants now incorporate stimuli-responsive materials and microelectronic components. Smart implants can be engineered to respond to external stimuli, such as temperature or pH, or be remotely controlled, allowing patients or healthcare providers to adjust drug release as needed. These electronic-based systems, however, present challenges, including higher production costs and a lack of biodegradability.
In addition to novel LADDS technologies, advances in manufacturing techniques, particularly 3D-printing, are expanding the potential of personalised drug delivery. This technology enables the production of intricate solid implants tailored to individual patient needs, allowing for more effective and customised treatments. Moreover, it enables the production of medical devices with complex geometries and intricate designs that are challenging to manufacture using conventional fabrication techniques. 3D-printing opens a range of possibilities for researchers and industry professionals in the development of LADDS. This technology can be used not only to create drug delivery systems tailored to patients' needs at the point of care but also to accelerate medical device development by streamlining the prototyping stage. Additionally, the cost of 3D printing equipment has decreased significantly in recent years, making this technology more accessible, including in developing countries.
As research continues, these emerging innovations are set to enhance the efficiency, accessibility, and precision of LADDS, offering improved therapeutic outcomes across a range of medical applications.
References
1. Holm R, Lee RW, Glassco J, DiFranco N, Bao Q, Burgess DJ, et al. Long-Acting Injectable Aqueous Suspensions—Summary From an AAPS Workshop. AAPS J [Internet]. 2023 Apr 28;25(3):49. Available from: https://link.springer.com/10.1208/s12248-023-00811-8
2. Affandi B, Santoso SSI, Djajadilaga, Hadisaputra W, Moeloek FA, Prihartono J, et al. Five-year experience with Norplant®. Contraception [Internet]. 1987 Oct;36(4):417–28. Available from: https://linkinghub.elsevier.com/retrieve/pii/0010782487900904
3. Friend DR. Advances in vaginal drug delivery. Drug Deliv Transl Res [Internet]. 2011 Jun 10;1(3):183–4. Available from: http://link.springer.com/10.1007/s13346-011-0030-6
4. Wu L, Janagam DR, Mandrell TD, Johnson JR, Lowe TL. Long-Acting Injectable Hormonal Dosage Forms for Contraception. Pharm Res [Internet]. 2015 Jul 22;32(7):2180–91. Available from: http://link.springer.com/10.1007/s11095-015-1686-2
5. Baum MM, Butkyavichene I, Gilman J, Kennedy S, Kopin E, Malone AM, et al. An Intravaginal Ring for the Simultaneous Delivery of Multiple Drugs. J Pharm Sci [Internet]. 2012 Aug;101(8):2833–43. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022354915314908
6. Goldspiel BR, Kohler DR. Goserelin Acetate Implant: A Depot Luteinizing Hormone-Releasing Hormone Analog for Advanced Prostate Cancer. DICP [Internet]. 1991 Jul 4;25(7–8):796–804. Available from: http://journals.sagepub.com/doi/10.1177/106002809102500716
7. Hamm R, Patel B, Whittlestone T, Persad R. Patient self-injection: a new approach to administering luteinizing hormone-releasing hormone analogues. BJU Int [Internet]. 2001 Dec 24;86(7):840–2. Available from: http://doi.wiley.com/10.1046/j.1464-410x.2000.00910.x
8. Schlegel P. A review of the pharmacokinetic and pharmacological properties of a once-yearly administered histrelin acetate implant in the treatment of prostate cancer. BJU Int [Internet]. 2009 Mar;103:7–13. Available from: http://doi.wiley.com/10.1111/j.1464-410X.2009.08383.x
9. Westphal M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC, et al. A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro Oncol [Internet]. 2003 Apr 1;5(2):79–88. Available from: http://academic.oup.com/neuro-oncology/article/5/2/79/1070081/A-phase-3-trial-of-local-chemotherapy-with
10. Elstad NL, Fowers KD. OncoGel (ReGel/paclitaxel) — Clinical applications for a novel paclitaxel delivery system. Adv Drug Deliv Rev [Internet]. 2009 Aug;61(10):785–94. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0169409X09001422
11. Daneshmand S, Pohar KS, Steinberg GD, Aron M, Cutie C. Effect of GemRIS (gemcitabine-releasing intravesical system, TAR-200) on antitumor activity in muscle-invasive bladder cancer (MIBC). J Clin Oncol [Internet]. 2017 May 20;35(15_suppl):e16000–e16000. Available from: http://ascopubs.org/doi/10.1200/JCO.2017.35.15_suppl.e16000
12. Macoul KL, Pavan-Langston D. Pilocarpine Ocusert System for Sustained Control of Ocular Hypertension. Arch Ophthalmol [Internet]. 1975 Aug 1;93(8):587–90. Available from: http://archopht.jamanetwork.com/article.aspx?articleid=631604
13. Haghjou N, Soheilian M, Abdekhodaie MJ. Sustained release intraocular drug delivery devices for treatment of uveitis. J Ophthalmic Vis Res [Internet]. 2011 Oct;6(4):317–29. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22454753
14. Wong IB, Teoh SC, Yeoh AE, Lingam G. Sustained-release ganciclovir implant as prophylaxis for cytomegalovirus retinitis in a child undergoing bone marrow transplantation. Eye [Internet]. 2013 Jul 26;27(7):890–1. Available from: http://www.nature.com/articles/eye201381
15. Margolis DA, Gonzalez-Garcia J, Stellbrink H-J, Eron JJ, Yazdanpanah Y, Podzamczer D, et al. Long-acting intramuscular cabotegravir and rilpivirine in adults with HIV-1 infection (LATTE-2): 96-week results of a randomised, open-label, phase 2b, non-inferiority trial. Lancet [Internet]. 2017 Sep;390(10101):1499–510. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0140673617319177
16. Grossman SA, Roberts N. Analgesic applications for a subcutaneous implant that continuously releases hydromorphone. Eur J Pain Suppl [Internet]. 2011 Nov;5(S2):439–42. Available from: http://doi.wiley.com/10.1016/j.eujps.2011.08.008
17. Nickel JC, Jain P, Shore N, Anderson J, Giesing D, Lee H, et al. Continuous Intravesical Lidocaine Treatment for Interstitial Cystitis/Bladder Pain Syndrome: Safety and Efficacy of a New Drug Delivery Device. Sci Transl Med [Internet]. 2012 Jul 18;4(143):143ra100-143ra100. Available from: https://stm.sciencemag.org/lookup/doi/10.1126/scitranslmed.3003804
18. Itzoe M, Guarnieri M. New developments in managing opioid addiction: impact of a subdermal buprenorphine implant. Drug Des Devel Ther [Internet]. 2017 May;Volume 11:1429–37. Available from: https://www.dovepress.com/new-developments-in-managing-opioid-addiction-impact-of-a-subdermal-bu-peer-reviewed-article-DDDT
19. Mohtashami Z, Esmaili Z, Vakilinezhad MA, Seyedjafari E, Akbari Javar H. Pharmaceutical implants: classification, limitations and therapeutic applications. Pharm Dev Technol [Internet]. 2020 Jan 2;25(1):116–32. Available from: https://www.tandfonline.com/doi/full/10.1080/10837450.2019.1682607
20. Dammerman R, Kim S, Adera M, Schwarz A. Pharmacokinetics and Safety of Risperidone Subcutaneous Implants in Stable Patients With Schizophrenia. Clin Pharmacol Drug Dev [Internet]. 2018 Mar;7(3):298–310. Available from: http://doi.wiley.com/10.1002/cpdd.428
21. Anselmo AC, Mitragotri S. An overview of clinical and commercial impact of drug delivery systems. J Control Release [Internet]. 2014 Sep;190:15–28. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0168365914002193
22. McGowan J, Taylor M. Paliperidone palmitate: monthly depot injection for schizophrenia. Prescriber [Internet]. 2012 Mar 5;23(5):15–9. Available from: http://doi.wiley.com/10.1002/psb.873
23. Stolovitzky JP, Kern RC, Han JK, Forwith KD, Ow RA, Wright SK, et al. In-office Placement of Mometasone Furoate Sinus Implants for Recurrent Nasal Polyps: A Pooled Analysis. Am J Rhinol Allergy [Internet]. 2019 Sep 23;33(5):545–58. Available from: http://journals.sagepub.com/doi/10.1177/1945892419850924
24. Parikh A, Anand U, Ugwu MC, Feridooni T, Massoud E, Agu RU. Drug-eluting nasal implants: formulation, characterization, clinical applications and challenges. Pharmaceutics [Internet]. 2014 May 27;6(2):249–67. Available from: http://www.mdpi.com/1999-4923/6/2/249
25. Cook DM, Biller BMK, Vance ML, Hoffman AR, Phillips LS, Ford KM, et al. The Pharmacokinetic and Pharmacodynamic Characteristics of a Long-Acting Growth Hormone (GH) Preparation (Nutropin Depot) in GH-Deficient Adults. J Clin Endocrinol Metab [Internet]. 2002 Oct;87(10):4508–14. Available from: https://academic.oup.com/jcem/article-lookup/doi/10.1210/jc.2002-020480
26. Chanson P, Boerlin V, Ajzenberg C, Bachelot Y, Benito P, Bringer J, et al. Comparison of octreotide acetate LAR and lanreotide SR in patients with acromegaly. Clin Endocrinol (Oxf) [Internet]. 2000 Nov 17;53(5):577–86. Available from: http://doi.wiley.com/10.1046/j.1365-2265.2000.01134.x
27. Johnson BA. Naltrexone long-acting formulation in the treatment of alcohol dependence. Ther Clin Risk Manag [Internet]. 2007 Oct;3(5):741–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18472999
28. Ho D-K, LeGuyader C, Srinivasan S, Roy D, Vlaskin V, Chavas TEJ, et al. Fully synthetic injectable depots with high drug content and tunable pharmacokinetics for long-acting drug delivery. J Control Release [Internet]. 2021 Jan;329:257–69. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0168365920306805
29. Coulter SM, Pentlavalli S, An Y, Vora LK, Cross ER, Moore J V., et al. In Situ Forming, Enzyme-Responsive Peptoid-Peptide Hydrogels: An Advanced Long-Acting Injectable Drug Delivery System. J Am Chem Soc [Internet]. 2024 Aug 7;146(31):21401–16. Available from: https://pubs.acs.org/doi/10.1021/jacs.4c03751
30. MeltPrep. What is VCM Technology? [Internet]. 2023 [cited 2023 Aug 30]. Available from: https://www.meltprep.com/vcm
31. Li L, Permana AD, Domínguez‐Robles J, Amir MN, Habibie H, Anjani QK, et al. Reservoir‐Type Subcutaneous Implantable Devices Containing Porous Rate Controlling Membranes for Sustained Delivery of Risperidone. Adv Healthc Mater [Internet]. 2025 Jan 16; Available from: https://onlinelibrary.wiley.com/doi/10.1002/adhm.202403689
32. Chua CYX, Jain P, Ballerini A, Bruno G, Hood RL, Gupte M, et al. Transcutaneously refillable nanofluidic implant achieves sustained level of tenofovir diphosphate for HIV pre-exposure prophylaxis. J Control Release [Internet]. 2018 Sep;286:315–25. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0168365918304711
33. Pons-Faudoa FP, Sizovs A, Di Trani N, Paez-Mayorga J, Bruno G, Rhudy J, et al. 2-Hydroxypropyl-β-cyclodextrin-enhanced pharmacokinetics of cabotegravir from a nanofluidic implant for HIV pre-exposure prophylaxis. J Control Release. 2019;306(March):89–96.
34. Delpor. DLP-160 [Internet]. 2019 [cited 2022 Apr 8]. Available from: https://delpor.com/products/dlp-160/
35. Tekko IA, Vora LK, Volpe‐Zanutto F, Moffatt K, Jarrahian C, McCarthy HO, et al. Novel Bilayer Microarray Patch‐Assisted Long‐Acting Micro‐Depot Cabotegravir Intradermal Delivery for HIV Pre‐Exposure Prophylaxis. Adv Funct Mater [Internet]. 2022 Feb 27;32(9):2106999. Available from: https://onlinelibrary.wiley.com/doi/10.1002/adfm.202106999
36. Peng K, Vora LK, Domínguez-Robles J, Naser YA, Li M, Larrañeta E, et al. Hydrogel-forming microneedles for rapid and efficient skin deposition of controlled release tip-implants. Mater Sci Eng C. 2021 Aug;127:112226.
37. aVaxziPen [Internet]. [cited 2025 Jan 29]. Available from: https://www.avaxzipen.com/








