Pharmaceutical Research and Development
The Power of Collaborations
Om Shelke, Chief Scientist, Sinomune Pharmaceutical Co. Ltd.
Collaboration in pharmaceutical R&D is pivotal for accelerating drug development, reducing costs, and fostering innovation. This article explores the role of cross-functional teamwork, the benefits of effective collaborations, and the challenges of poor coordination. Highlighting real-world examples like Pfizer-BioNTech and Merck-AstraZeneca, it emphasizes strategies such as clear communication, integrated project management, and cultural alignment to enhance R&D efficiency. By fostering collaboration, the pharmaceutical industry can overcome challenges, streamline processes, and deliver life-saving therapies to market faster.
1. Introduction
Collaboration is a process of aligning the goals of multiple people or departments or companies for a common project or firm output. Collaboration in pharmaceutical R&D could occur among multiple departments of the same or different company, between two pharmaceutical firms, or, between contract research organizations and contract manufacturing operations. Effective collaboration among business development, formulation development, analytical development, manufacturing technology, clinical department, regulatory affairs, and marketing teams is essential for successful product launches in pharmaceutical companies.
Pharmaceutical R&D is a high-stakes endeavour that involves the discovery of new drugs, drug product development, preclinical testing, manufacturing, clinical trials, regulatory approvals, and post-market surveillance. The process is time-consuming, expensive, and fraught with risks, as only a small percentage of drug candidates make it to market. In recent years, the industry has witnessed a shift from in-house R&D to collaborative models, including partnerships between pharmaceutical companies, academic institutions, contract research organizations (CROs), and biotechnology firms. These collaborations are driven by the need to share expertise, reduce costs, and accelerate the development of life-saving therapies.
In highly dynamic cutting-edge technology pharmaceutical industries the business firm faces rapidly transforming environments with enormous vying and perpetually modifying technologies as well as market needs. Effective collaborations create a new market opportunity and encourage fast product solutions. Resources could be rapidly activated in exigencies to adapt to new market demands to transform knowledge into innovative products and technological improvements in the most effective way through collaboration.
This article delves into the importance of collaborations in pharma R&D, the role of cross-functional departments, the positive and negative impacts of collaboration, and strategies to enhance project efficiency through effective teamwork.
2. Research and Development in Pharma: An Overview
2.1 The R&D Process
The pharmaceutical R&D process can be divided into several key stages starting from drug discovery, followed by preclinical research, formulation design, manufacturing, clinical trials, regulatory approvals, and ending with post-marketing surveillance. Drug discovery involves the screening of millions of molecules and the identification of potential drug targets and compounds. Preclinical research involves the testing of lead compounds either in-vitro or animal models to assess safety and efficacy for target indication. Formulation design involves the conversion of drug powder into a suitable stable dosage form at R&D followed by the technology transfer to the commercial site for manufacturing of bulk clinical batches. Human clinical trials are conducted in three phases to evaluate safety, dosage, and effectiveness. The entire dossier is compiled together and submitted the data to regulatory agencies (e.g., FDA, EMA) for marketing authorization of drug approval. The research for pharmaceutical R&D doesn’t end with product marketing approval, further, it involves post-market surveillance to monitor the drug's safety and efficacy after commercialization.
2.2 Challenges in Pharma R&D
There are numerous challenges in pharmaceutical R&D and major challenges are high cost, long timelines, high failure rates, regulatory changes, and regulatory requirements. The average cost of developing a new drug is estimated at $2.6 billion which is very high. Additionally, the timeline for the pharmaceutical drug production development is 10-15 years to bring a new drug to market. The failure rates are very high in pharmaceutical R&D especially for new chemical entities and only about 12% of drug candidates that enter clinical trials are approved. Stringent regulatory requirements make the drug approval process even more time-consuming. The regulatory landscape is very large and changing constantly to ensure the complete safety of drug products before entering the market. Delay approvals given these challenges, collaborations have become essential to streamline R&D processes and improve outcomes.
3. Cross-Functional Departments in Pharma R&D
Pharma R&D involves multiple cross-functional departments, each playing a critical role in the drug development process. Effective collaboration between these departments is crucial for success. The key departments in R&D involve discovery research, preclinical development, clinical development, regulatory affairs, technology transfer, manufacturing, quality assurance, marketing, and commercialization. Each department has a unique role in pharmaceutical development and the project management team co-ordinate between all of them for proper functioning and alignment towards a final delivery.
Cross-functional collaboration is paramount for the proper functioning of all the operations in the R&D. Pharmaceutical R&D is an integration expert having diverse skills and knowledge to address complex challenges. Faster and correct decision-making reduces delays by facilitating communication and coordination. Identifies potential issues early and develops contingency plans to mitigate the potential risks.
4. Positive Impact of Effective Collaborations
Effective collaborations in pharma R&D can lead to significant benefits, including accelerated timelines, reduced costs, and improved outcomes. Innovation usually demands time-consuming exploratory strategies; urgent times provoke business firms to swiftly utilize their existing resources for immediate action.
Innovation is accelerated by knowledge sharing through effective collaborations which enables the sharing of expertise and resources, leading to faster discovery and development. Partnerships with academic institutions and biotech firms provide access to cutting-edge technologies and save the timelines for development. Effective collaborations lead to cost efficiency through resource and risk sharing. Collaborations between two different firms reduce the need for redundant investments in infrastructure and personnel. For example, extensive collaboration is needed during clinical study which is highly important for product approval and is outsourced as each company doesn’t maintain a clinical department to save manpower and cost. Partnerships distribute financial risks among stakeholders. The small business firms specialized in particular areas are burgeoning in the 21st century. This is leading to increased collaboration with the experts of defined specialty rather than shooting in the dark. For instance, Foamix is specialized only in foam technology and products.
Problem-solving is enhanced by the pooling of diverse expertise and knowledge and by the use of collaborative teams to solve complex product problems. Different team members bring unique skills and insights, enabling the team to examine problems from different perspectives and develop comprehensive solutions.
The research outcomes could be enhanced significantly by maintaining the high-quality product through collaborative efforts leading to better-designed studies and higher-quality data. For instance, while planning the development, one expert may not foresee the potential problem but the other expert has faced the problem and has solutions for the potential problems. So, this will help to avoid unforeseen problems and ready solution for the same. Effective collaboration and communication with regulatory agencies speed up the drug product approval process.
Collaboration fosters a sense of togetherness and common purpose within a team. When employees feel valued and included in the decision-making process, they are more engaged in their work, leading to greater job satisfaction and overall productivity.
The epic collaboration between Pfizer and BioNTech worked in a collaboration that resulted in the rapid development and approval of a highly effective vaccine Pfizer-BioNTech COVID-19 Vaccine. Another example of effective collaboration is the Merck-Keystone Partnership for a successful collaboration in cancer immunotherapy.
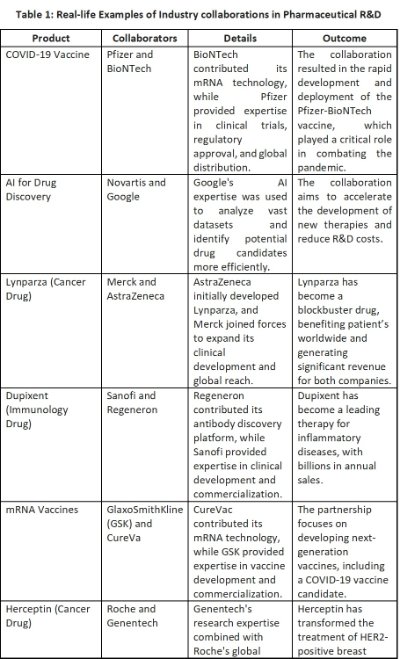
5. Negative Impact of Poor Collaboration
While collaborations offer numerous benefits, miscommunication and poor coordination can have detrimental effects on R&D projects. The major impact is delays in timelines, a lack of collaboration leads to poor communication which creates conflicts in priorities. Bureaucratic hurdles in complex organizational structures can slow down decision-making.
Poor collaboration in R&D increases the costs due to rework, repetition of work, and resource wastage. Miscommunication can result in errors that require costly corrections through reworking. Inefficient use of resources due to poor coordination increase resource wastage.
Another major impact of poor collaboration is project failures due to regulatory rejections or clinical failures. Incomplete or inaccurate data submissions to regulatory agencies could lead to regulatory rejections at the initial stages. Poorly designed clinical trials due to poor collaboration could result in trial failures which is deadliest for the company and project.
The second French Community Innovation Survey (CIS) indicates that 14% of R&D collaborating firms had to abandon or delay their innovation projects due to difficulties in their partnerships, an outcome which we term “cooperation failures”.
6. Strategies to Enhance Project Efficiency through Effective Collaboration
To maximize the benefits of collaborations and minimize the risks, pharma companies must adopt strategies to improve communication and coordination.
Establish a clear communication channel for regular meetings and unified platforms for effective collaboration. The scheduled regular meetings help to ensure clear alignment, avoid confusion, increase understanding, provide clear updated timelines, increase visibility, and address issues promptly. The use of collaborative tools and platforms to facilitate real-time communication by reducing barriers.
The defined roles and responsibilities provide the role clarity and hold accountable to the individual or department for completing the task within a stipulated time. The role clarity should be clearly defined for each team member and mechanisms should be established to hold team members accountable for their tasks. This led to the important aspects of ownership through which success is driven by the individuals. Ownership is much needed for task completion which could only be given through collaborative work fashion.
The paramount department to improve the collaboration is Integrated Project Management. The Integrated Project Management department develops, monitors, and updates detailed project plans with clear milestones and timelines. Additionally, they identify potential risks, consult for expert opinion, and develop mitigation strategies. Furthermore, they arrange all the logistics such as materials, manpower, or information required for successful project deliveries through effective collaborations
The culture of the organization matters a lot for effective collaboration and crossing the boundaries of thoughts. The organization's culture could either kill or nurture the product effectively. Cultural Alignment is really important for effective collaboration. Fostering a culture of collaboration by aligning teams around a shared vision and goals expedite the project delivery. Trust Building in the organization for each individual is paramount which encourages open communication to share positive or negative thoughts for common goals. It could be done in multiple ways but the most preferred is trust-building activities.
Another important aspect of effective collaboration is data sharing and leveraging the latest technology for the team members. Data sharing through cloud-based platforms provides seamless data sharing and collaboration which enhances the clarity of tasks or goals. Leveraging artificial intelligence and data Analytical tools helps to optimize decision-making.
7. Conclusion
Collaborations are a driving force behind innovation and efficiency in pharma R&D. By bringing together diverse expertise and resources, collaborations enable faster, cost-effective, and high-quality drug development. However, the success of these collaborations depends on effective communication, clear roles, and integrated project management. Pharma companies must prioritize collaboration strategies to overcome challenges, mitigate risks, and achieve their R&D goals. As the industry continues to evolve, fostering a culture of collaboration will be key to addressing unmet medical needs and improving global health outcomes.
References
1. DiMasi, J. A., Grabowski, H. G., & Hansen, R. W. (2016). Innovation in the pharmaceutical industry: New estimates of R&D costs. Journal of Health Economics, 47, 20-33.
2. Paul, S. M., Mytelka, D. S., Dunwiddie, C. T., Persinger, C. C., Munos, B. H., Lindborg, S. R., & Schacht, A. L. (2010). How to improve R&D productivity: The pharmaceutical industry's grand challenge. Nature Reviews Drug Discovery, 9(3), 203-214.
3. Munos, B. (2009). Lessons from 60 years of pharmaceutical innovation. Nature Reviews Drug Discovery, 8(12), 959-968.
4. Pisano, G. P. (2006). Science Business: The Promise, the Reality, and the Future of Biotech. Harvard Business Press.
5. Hughes, B. (2010). Pharma pursues novel models for academic collaboration. Nature Reviews Drug Discovery, 9(11), 837-838.







